12 Essential Q&As for Interpreting Cyclic Voltammetry (CV) in Battery Research
Source: WeChat Official Account “Learn Batteries Together” 来源于微信公众号 一起学电池
1. Q: What system is used for Cyclic Voltammetry (CV) measurements, and why?
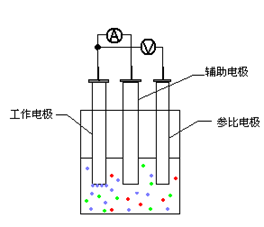
A: CV is a method used to characterize electrode reaction parameters. An electrode reaction typically consists of a redox system, a supporting electrolyte, and an electrode assembly. In the vast majority of cyclic voltammetry measurements, a three-electrode system is utilized.
The reason for using a three-electrode system is that during the polarization process, the potentials of both the working electrode and the counter (auxiliary) electrode are constantly changing. Furthermore, the ohmic potential drop (IR drop) generated by the polarization current passing through the solution between these electrodes would be added to the measured electrode potential. This would interfere with the accuracy of the results; a three-electrode setup mitigates these effects by separating the current-carrying circuit from the potential-sensing circuit.
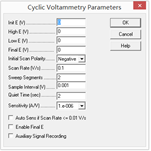
2. Q: What are the critical parameters in a Cyclic Voltammetry (CV) test? How should these parameters be selected during measurement?
A: When performing CV measurements, several key parameters must be configured. Selecting these values depends on the redox characteristics of the target substance and the specific objectives of your research.
The essential parameters include:
Initial Potential: The starting voltage of the scan.
Upper Limit Potential: The highest voltage point of the triangle wave.
Lower Limit Potential: The lowest voltage point of the triangle wave.
Initial Scan Direction: Whether the scan begins moving toward the anodic (positive) or cathodic (negative) direction.
Scan Rate ($v$): The speed of the potential change (e.g., 1 mV/s).
Number of Segments: Two segments usually constitute one full cycle (1.0 cycle).
Sampling Interval: The potential step between recorded data points.
Rest Time: The equilibration period before the scan begins.
Sensitivity (Current Range): Should be set at the same order of magnitude as the expected current (or one level higher). It should be as low as possible to ensure resolution, provided that “Overflow” does not appear on the interface.
Selection Strategy: Usually, an initial “scouting” scan is performed over a wide potential range to identify the locations of the redox peaks. Once the peaks are found, the potential window is narrowed to a more suitable range. Note: Selecting a potential range that is too wide may trigger the oxidation or reduction of the solvent (e.g., water splitting), which can mask the peaks of the target substance.
3. Q: How do you identify and determine the redox peaks in a Cyclic Voltammetry (CV) plot?
A: In a cyclic voltammetry (CV) curve, the reduction peak corresponds to the cathodic reaction. The current measured here is the cathodic current. A more positive peak potential (Epc) and a higher peak current (ipc) indicate that the substance is more easily reduced. Conversely, the oxidation peak corresponds to the anodic reaction, and its current is the anodic current. A more negative peak potential (Epa) and a higher peak current (ipa) indicate that the substance is more easily oxidized.
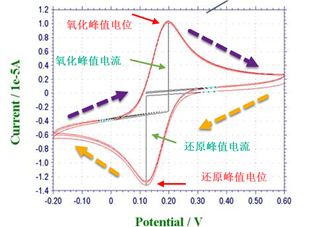
① Judging by potential, for the same redox couple, the oxidation peak is usually located at a more positive potential than the reduction peak. That is to say, the peak with the more positive peak potential is the oxidation peak, and the peak with the more negative peak potential is the reduction peak; this is a result of polarization.
② Scanning direction: In the parameter settings of cyclic voltammetry, there is an initial scan polarity (negative or positive). A positive scan (scanning from low voltage to high voltage) refers to moving from a negative potential to a positive potential, and the resulting peak is the oxidation peak. A negative scan (scanning from high voltage to low voltage) refers to moving from a positive potential to a negative potential, indicating that the external circuit is adding electrons to the electrode. Ions in the solution that undergo reduction easily (e.g., ferric ions) approach the electrode and gain electrons, causing a reduction reaction to occur; therefore, the resulting peak is the reduction peak. Thus, the more negative the potential, the stronger the tendency for reduction, and the resulting peak is the reduction peak.
Scanning in the positive or negative direction at the same voltage will result in oxidation and reduction, respectively; it is the scanning direction, rather than the voltage level itself, that determines whether oxidation or reduction occurs.
4. Q: Which parameters need to be analyzed in a Cyclic Voltammetry (CV) curve?
A: Several important parameters that can be obtained from a cyclic voltammogram are: anodic peak current (ipa), cathodic peak current (ipc), anodic peak potential (Epa), and cathodic peak potential (Epc).
The method for measuring and determining the peak current ip is: extrapolate a tangent line along the baseline to below the peak, and draw a vertical line from the peak tip to the tangent line; the height of this segment is ip. Ep can be read directly from the corresponding point on the horizontal axis at the peak tip.
5. Q: How to use Cyclic Voltammetry (CV) to determine the reaction mechanism?
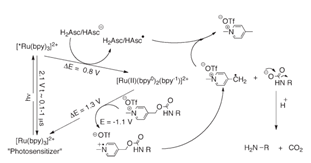
A: Taking the photodegradation reaction in the figure below as an example to speculate on its reaction process: ruthenium bipyridine (Ru(bpy)3 2+) acts as the oxidant, and ascorbic acid acts as the reductant.
The relationship between activation energy and potential is as follows:
By combining factors such as solvent coefficients, we can obtain the Gibbs free energy calculation formulas for two different reaction processes:
Substituting the potential value into the equation yields the Gibbs free energy, and the reaction mechanism can be determined based on the sign of the free energy.
6. Q: How to use Cyclic Voltammetry (CV) to determine reaction characteristics?
A: ① Use cyclic voltammetry to determine if a reaction is reversible (generally, these two conditions are sufficient). a. The absolute value of the ratio of the anodic peak current to the cathodic peak current equals 1. [Sometimes for the same system, different scan rates will affect its reversibility to some extent. Generally speaking, scan rate has no effect on the peak potential, but the higher the scan rate, the higher the electrochemical reaction current.] b. The potential difference between the anodic and cathodic peaks is approximately 59/n (mV), where n is the number of electrons transferred (usually at a temperature of 293K). [However, since our experiments are generally not conducted at this exact temperature, using this value involves some error; generally, it is considered a reasonable error as long as the value is kept below 100 mV.]
② Determine whether it is a diffusion-controlled or adsorption-controlled reaction: Change the scan rate and observe whether the peak current is proportional to the scan rate or its square root. a. If there is a linear relationship with the scan rate, it is surface-controlled (adsorption). b. If there is a linear relationship with the square root of the scan rate, it is diffusion-controlled.
Several Questions Regarding Cyclic Voltammetry (CV)
1. Why is a reference electrode necessary?
When the potential difference of the working electrode relative to the reference electrode is known, the potential difference at the interface between the electrode and the solution is indirectly known. Therefore, by controlling the potential difference of the working electrode relative to the reference electrode, we indirectly control the potential difference at the electrode-solution interface.
2. What should be noted when using a three-electrode electrolytic cell?
When performing cyclic voltammetry in an electrolytic cell, the distance between the working electrode and the counter electrode should be kept constant. The distance between the reference electrode and the working electrode should be as close as possible. The working electrode, counter electrode, and reference electrode must remain fixed in position.
3. Which is the oxidation peak and which is the reduction peak?
The peak at the higher potential is the oxidation peak; the peak at the lower potential is the reduction peak.
4. Why are some peaks incomplete?
One reason is that the potential range is too narrow; the oxidation reaction has not been fully completed before the reverse scan begins. Another reason is that the scan rate is too high, causing the oxidation peak to shift significantly toward a more positive potential, which results in an incomplete peak within the same potential range.
5. How is the scan rate determined?
If the goal is simply to investigate the redox reactions occurring on the electrode, the scan rate should be less than 1 mV/s. At this rate, the electrode surface can be regarded as a steady-state process. If you want to examine the proportion of non-Faradaic current simultaneously, the scan rate can be increased. In this case, the peak positions will shift due to the internal resistance of the electrode, the rectangular portion of the curve will increase, and some redox peaks may be masked.
6. Why does the electrolyte change color during cyclic voltammetry in an electrolytic cell?
This is due to the redox reactions occurring on the electrode. If the substance on the electrode surface is not firmly attached, ions will dissolve into the electrolyte, causing the electrolyte to change color.
Of course, performing cyclic voltammetry tests using an electrochemical workstation is quite simple. The most important part is the data analysis, which allows the cyclic voltammetry test to be used to its fullest potential.
Due to the limited knowledge and English level is inevitable errors and omissions, if there are errors or infringement of the text, please contact me as soon as possible by private letter, I will immediately be corrected or deleted.