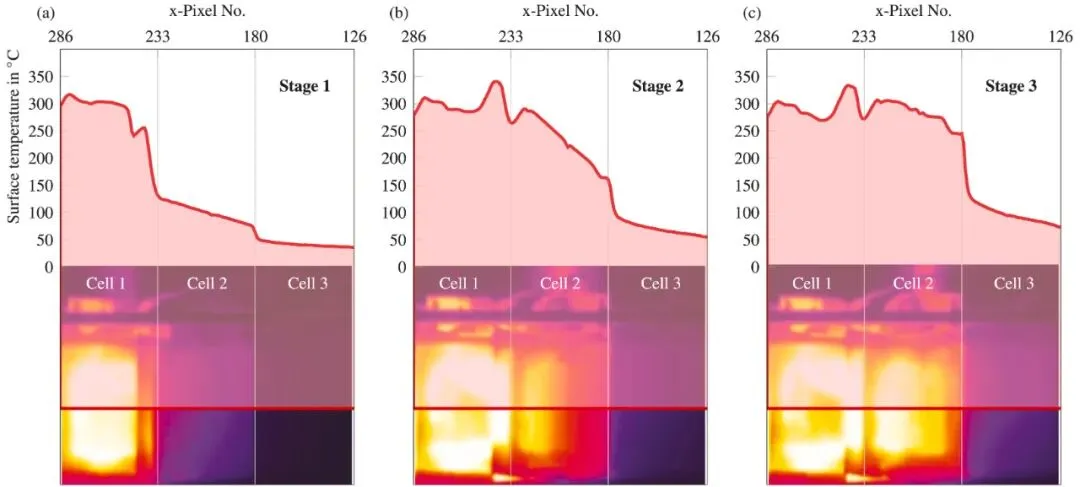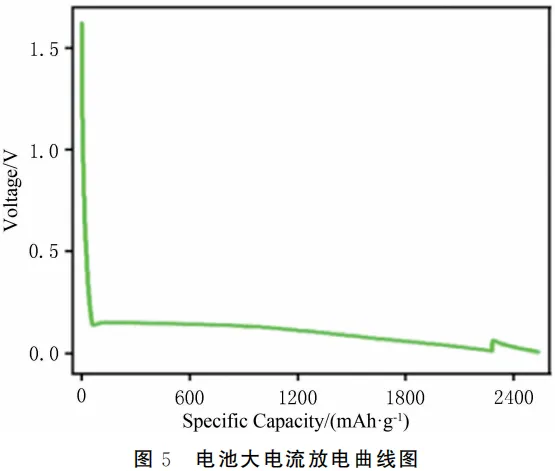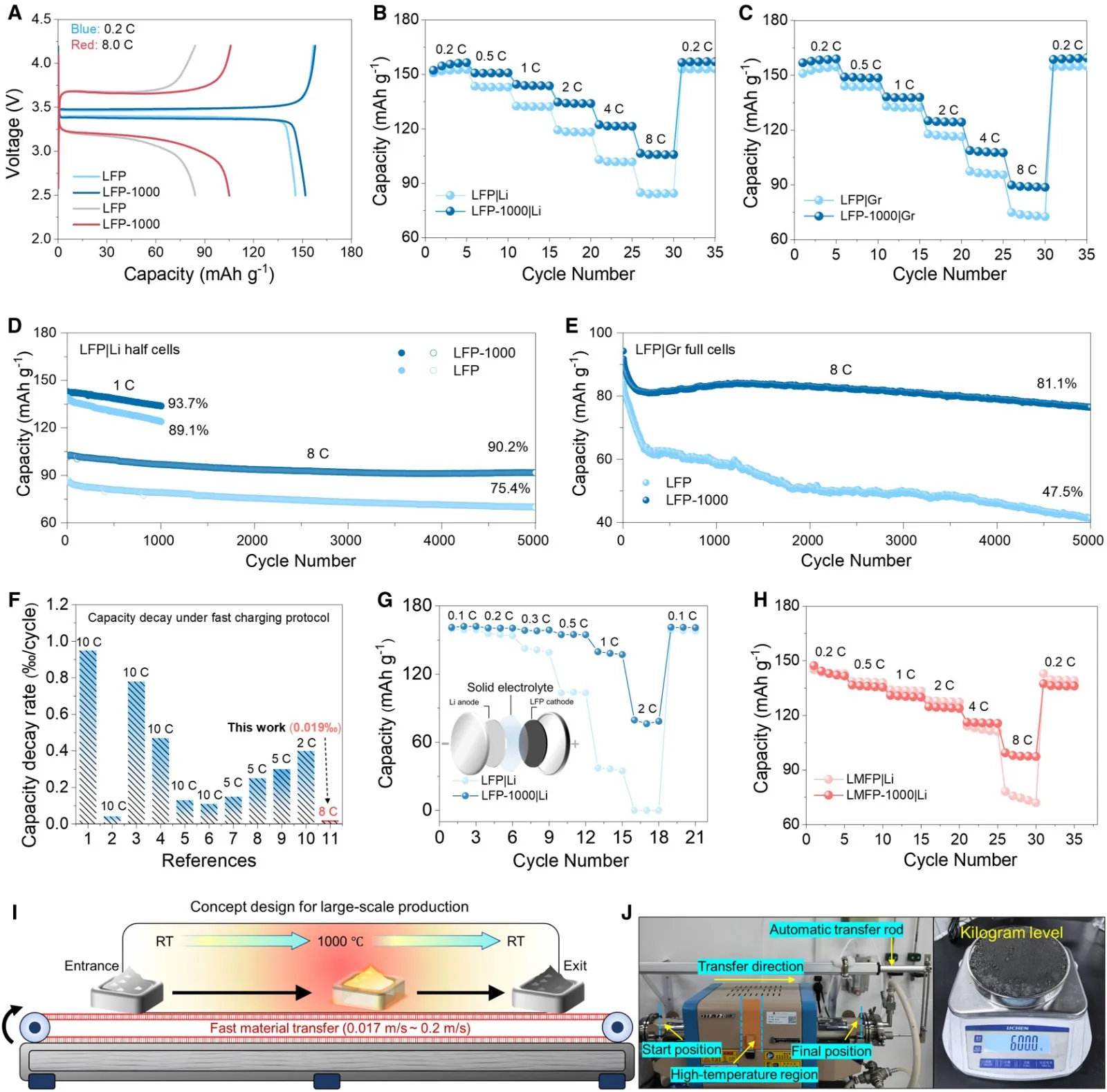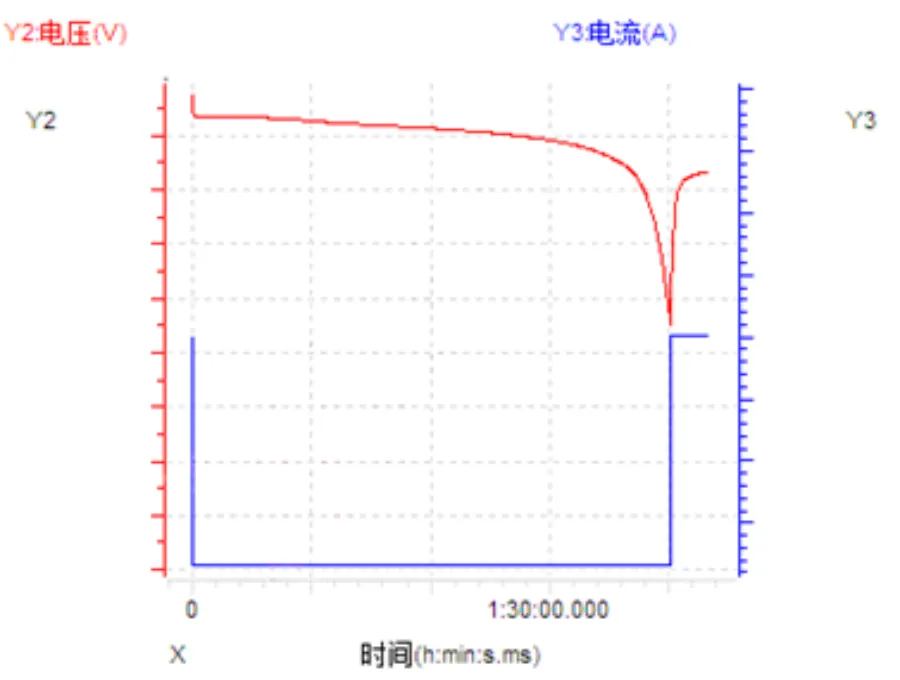
How to evaluate and improve the electrolyte wettability of lithium-ion batteries? 2026
How to evaluate and improve the electrolyte wettability of lithium-ion batteries? First of all, let us understand the factors that affect electrolyte wettability. Factors Affecting Electrolyte Wettability of lithium-ion batteries The essence of electrolyte wettability is the synergistic effect of materials, processes, and structures. The uniformity of wetting can be systematically improved by reducing electrolyte viscosity (e.g., using low-viscosity solvents such as DEC/EMC), adding surfactants (fluorinated wetting agents can significantly reduce surface tension), optimizing electrode porosity (adjusting compaction density to avoid “dead zones”), and adopting vacuum-assisted heating injection processes. Electrolyte wettability directly determines ion transport efficiency, interfacial reaction uniformity, and overall battery performance. Poor wetting can lead to low active