Primary Battery: Technical Overview and Operational Principles
Definition and Functional Mechanism A primary battery, frequently categorized as a non-rechargeable galvanic cell, serves as a standalone energy source designed to convert chemical energy into electrical power through spontaneous electrochemical reactions. Unlike secondary cells, these units are engineered for a unidirectional discharge cycle, delivering a consistent current to an external load once the circuit is closed between its two polar terminals.
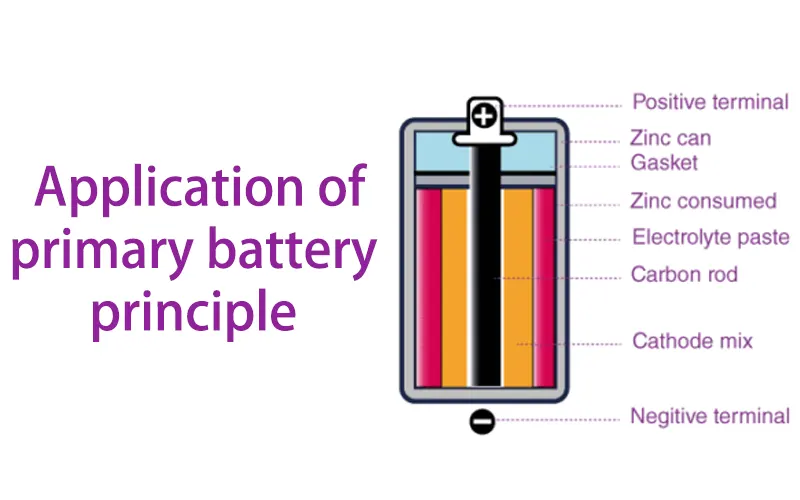
Core Components and Structural Composition The architecture of a primary cell typically integrates one or multiple high-purity chemical active materials. The fundamental electrochemical assembly comprises:
The Anode (Negative Electrode): The site where oxidation occurs, releasing electrons to the external circuit.
The Cathode (Positive Electrode): The site of reduction, receiving electrons from the load.
The Electrolyte Matrix: A critical conductive medium—available in either liquid (wet cell) or solid-state/gel (dry cell) formats—that facilitates ionic transport between the electrodes while preventing internal short-circuiting.
Operational Dynamics The performance of a primary battery is governed by the kinetics of its internal chemistry. As the reaction proceeds, the active materials are irreversibly consumed, defining the battery’s service life and energy density. This robust, maintenance-free design makes primary cells the preferred choice for applications requiring long-term reliability and immediate power readiness.
Electrochemical Discharge Dynamics and Lifecycle Constraints
The Mechanism of Galvanic Discharge Upon establishing a conductive path via an external load, the cell initiates a series of synchronized Redox (Reduction-Oxidation) kinetics. The operational efficiency of this process is driven by two simultaneous half-cell reactions:
Anodic Oxidation: The negative electrode serves as the electron donor, releasing charge carriers into the external circuit.
Cathodic Reduction: The positive electrode acts as the electron sink, consuming the migrated electrons to complete the electrochemical circuit. This regulated flow of electrons constitutes the operating current, providing the necessary wattage to power connected industrial or consumer-grade components.
Irreversibility and Service Life Termination A defining technical constraint of primary battery systems is their non-rechargeable architecture. Unlike secondary (rechargeable) chemistries, the internal active materials undergo an irreversible phase transformation or chemical exhaustion. Once the stoichiometric balance of the reactants is depleted or the internal resistance ($R_i$) reaches a critical threshold, the cell is deemed “spent.” At this stage, the electrochemical potential cannot be restored through external charging, necessitating a complete unit replacement to regain power functionality.
Positive electrode/Cathode
The Cathode: Electrochemical Potential and High-Voltage Reduction Kinetics
Fundamentals of Positive Electrode Polarity
The positive electrode, technically termed the cathode, operates as the higher-potential terminal within an electrochemical cell. In any galvanic system, the cathode is the site of reduction reactions, characterized by its ability to act as an electron sink. Its primary functional role is to facilitate the migration of charge carriers by attracting a steady flux of electrons from the negative electrode (anode) through the external load to maintain charge neutrality.
Structural Integration and Ionic Transport
The cathode is architecturally isolated from the anode by a conductive electrolyte matrix or a specialized separator, collectively forming the cell’s electrochemical core. Upon circuit closure, the active material within the cathode undergoes a reduction phase, accepting electrons and intercalating (or reacting with) ionic species. This process is critical for sustaining the battery’s electromotive force (EMF) and overall power output.
Material Selection and Performance Benchmarks
The strategic selection of cathode chemistries is the primary determinant of a battery’s energy profile. Different electrochemical systems utilize specialized active materials to achieve specific performance targets:
Zinc-Carbon Systems: Utilize high-purity carbon/manganese dioxide composites.
Lead-Acid Batteries: Employ lead dioxide (PbO2) structured with lead-calcium alloys for enhanced mechanical stability.
Advanced Lithium-ion (Li-ion) Platforms: Leverage high-capacity compounds such as LFP (Lithium Iron Phosphate), LCO (Lithium Cobalt Oxide), or NCM (Nickel Cobalt Manganese).
The chemical composition and crystalline structure of these materials directly dictate the cell’s nominal voltage, gravimetric energy density, and cycle-life stability.
Technical Summary
In essence, the cathode is defined by its high-voltage reduction kinetics and its role as the positive polarity plate. By acting as the counterpart to the anode, it drives the electron flow necessary for power delivery. The ongoing innovation in cathode material science remains the cornerstone for optimizing battery performance across consumer electronics and EV applications.
Negative electrode/Anode
The Anode: Low-Potential Oxidation and Electron Flux Dynamics
Functional Definition of Negative Polarity The negative electrode, or anode, represents the low-potential terminal in an electrochemical system. In the context of battery discharge, the anode is the fundamental site for oxidation reactions. Functioning as the electron donor, it establishes the negative polarity of the cell, facilitating the liberation of electrons which then migrate through the external circuit toward the cathode.
Electrochemical Architecture and Ionic Balance Within a standard cell configuration, the anode is strategically isolated from the cathode by an ion-permeable separator and immersed in a specialized electrolyte medium. Upon the engagement of an external load, the anode initiates a localized oxidation process. This reaction involves the loss of electrons from the active material, coupled with simultaneous interaction with ionic species to preserve the system’s internal charge equilibrium.
Material Engineering and Performance Optimization The chemical composition of the anode active material is a critical factor in determining the overall efficiency and energy density of the battery. Material selection varies significantly across different energy storage technologies:
Primary Zinc-Carbon Cells: Typically utilize high-purity metallic zinc.
Lead-Acid Systems: Employ porous lead (Pb) to maximize surface area and reactivity.
Secondary Lithium-ion (Li-ion) Platforms: Primarily rely on graphite or advanced silicon-carbon (Si-C) composites for improved capacity.
The specific electrochemical properties of the anode material directly influence the cell’s nominal voltage, rate capability, and cycle-life durability.
Technical Conclusion In summary, the anode is defined by its role as the source of electron flow during oxidation at a lower electrochemical potential. As the essential counterpart to the cathode, its material properties are pivotal in shaping the battery’s performance envelope, from discharge stability to long-term reliability in diverse applications.
Electrode
The electronic conductor or semiconductor in contact with an electrolyte solution or electrolyte
An electrode is an electronic conductor or semiconductor in direct contact with an electrolyte solution or electrolyte. Electrodes have wide applications in the field of electrochemistry, including energy storage devices, electrochemical sensors, electrolysis cells, and more. Let’s provide a detailed explanation of electrodes from a professional perspective and provide some practical examples.
Electrode Systems: Architecture, Functionality, and Cross-Domain Applications
Core Mechanism and Charge Transfer Electrodes serve as the fundamental conductive interfaces in electrochemical systems, providing the requisite pathways for electron migration between an external circuit and an electrolyte. In an electrolytic configuration, these components are classified based on the direction of current flow: the anode facilitates current entry into the electrolyte, whereas the cathode enables the transition of current back into the external circuitry.
Energy Storage Applications: Battery Electrodes
In galvanic systems (batteries), the electrodes are the primary sites for energy conversion. The negative electrode (the anode during discharge) typically utilizes high-capacity active materials—ranging from zinc in legacy zinc-carbon cells to high-purity graphite in modern Lithium-ion (Li-ion) batteries. Conversely, the positive electrode (the cathode during discharge) is comprised of reactive compounds such as LCO (Lithium Cobalt Oxide) or manganese-based structures. These materials facilitate the reversible redox (reduction-oxidation) kinetics, effectively storing or releasing electrical energy through the absorption and liberation of electrons.
Precision Sensing: The Working Electrode
Beyond power storage, electrodes are pivotal in electrochemical sensing technology. The “working electrode” acts as the sensory heart of the device, often fabricated from noble metals like gold (Au) and platinum (Pt), or advanced carbon allotropes like carbon nanotubes (CNTs). These materials are selected for their exceptional electrical conductivity and chemical inertness, allowing for the precise quantification of chemical concentrations and the monitoring of bio-molecular activities in complex environments.
Industrial Processing: Electrolysis and Electroplating
In industrial electrolytic cells—such as those used for aluminum smelting or electrodeposition—electrode selection is governed by the ability to withstand extreme operational stressors, including high current densities, thermal loads, and corrosive media. For instance, in aluminum electrolysis, carbon-based cathodes are paired with conductive iron or specialized anodes to endure the harsh reductive environment.
Optimization Factors in Electrode Design
The efficacy of any electrochemical application is intrinsically linked to the morphology and structural integrity of the electrode surface. Key design considerations include:
High Conductivity: To minimize Ohmic losses (IR drop).
Chemical Stability: To prevent premature degradation in aggressive electrolytes.
Electrocatalytic Activity: To lower the overpotential and accelerate reaction rates.
By optimizing surface porosity and material purity, engineers can significantly enhance the efficiency and lifespan of the entire electrochemical system.
Reference electrode
Reference Electrodes: The Standard for Precision Potential Measurement
Definition and Ideal Electrochemical Characteristics
In high-precision electrochemical analysis, a reference electrode (RE) serves as a constant potential datum point. Ideally, a reference electrode operates under conditions of near-zero polarization, maintaining a highly stable and well-defined electrochemical potential regardless of the experimental environment. Since it carries negligible current, it acts as a “calibrated ruler” against which the absolute potential of a working electrode (WE) can be accurately quantified.
The Role of Reference Potentials in Analysis
The primary utility of a reference electrode lies in its ability to decouple the potential of interest from the overall cell voltage. Unlike the working electrode, which undergoes dynamic surface reactions, the RE maintains a fixed interfacial potential. This stability is critical for:
Potentiostatic Control: Ensuring the intended voltage is applied precisely at the electrode-electrolyte interface.
Overpotential Analysis: Distinguishing between kinetic, Ohmic, and mass-transfer losses during battery cycling.
Standard Benchmarks and Practical Variants
Several established electrochemical couples are utilized to provide a reliable reference scale:
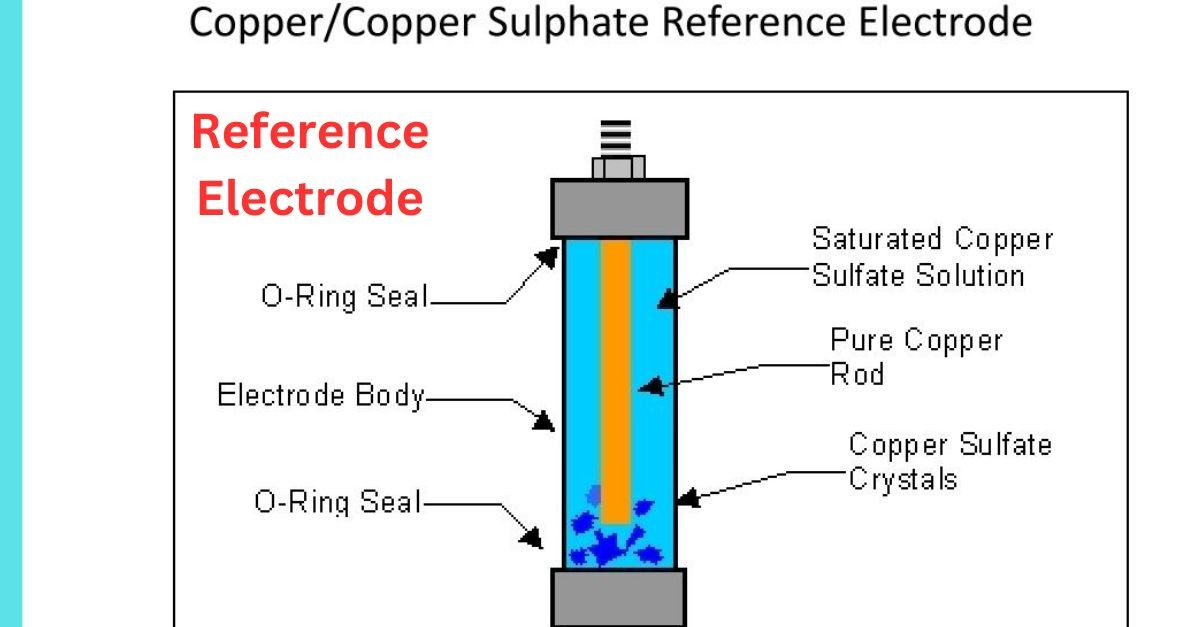
Standard Hydrogen Electrode (SHE): The primary thermodynamic benchmark, defined by the redox equilibrium of H2 gas at 1 atm on a platinum surface in an acidic medium, with its potential conventionally set to zero volts (0.000V).
Saturated Calomel Electrode (SCE): A secondary standard based on the Hg/Hg2Cl2 couple, favored in laboratory settings for its robust potential stability.
Silver/Silver Chloride Electrode (Ag/AgCl): Perhaps the most prevalent reference in modern battery research and bio-electrochemistry due to its compact design and minimal temperature coefficient.
Technical Significance in Battery R&D
In advanced battery testing (such as three-electrode cell configurations), the reference electrode allows researchers to monitor the individual health and degradation of the anode and cathode separately. By eliminating the interference of the counter electrode’s polarization, engineers can obtain high-fidelity data on material performance, which is essential for optimizing Solid Electrolyte Interphase (SEI) stability and long-term cycle life.
Practical Deployment and Selection Criteria for Reference Electrodes
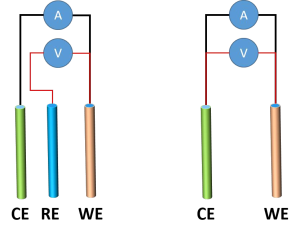
Implementation in Electrochemical Characterization In field applications and laboratory research, reference electrodes (RE) are utilized as the stable potential anchor within a three-electrode configuration. By pairing the RE with a working electrode (WE) and submerging both in a common electrolyte, researchers can employ a high-impedance voltmeter or potentiostat to capture the precise potential difference between them. This setup ensures that the measured voltage reflects the true interfacial kinetics of the WE, effectively isolating it from the polarization effects of the counter electrode.
Strategic Selection Based on Operational Windows The selection of an appropriate reference system is governed by the specific thermodynamic requirements of the application. Not all reference electrodes are interchangeable; the choice must align with the target electrochemical window:
High-Potential Analysis: For investigations into high-voltage redox reactions or advanced battery cathode testing, a reference electrode with a broader potential range—such as a pseudo-reference platinum (Pt) electrode—may be necessitated to prevent interference from the reference material’s own oxidation.
Compatibility with Media: The RE must also be chemically compatible with the electrolyte (e.g., non-aqueous vs. aqueous) to avoid junction potential errors or contamination.
Technical Summary To conclude, the reference electrode is a cornerstone of electrochemical precision, defined by its near-ideal non-polarization characteristics and consistent potential output under zero-current conditions. It provides the essential “zero-point” or datum for voltage quantification across diverse sectors. Whether in fundamental research, complex chemical analysis, or large-scale industrial battery manufacturing, the RE remains an indispensable tool for deciphering the intricate potential dynamics of electrochemical systems.
Learn more about NEWARE
Related News:
- Design, Assembly, and Testing of Full Coin Cells: Tutorials and Case Studies 2026 post
- 10 mins Guide to the Application and Customization of Three-Electrode Systems In Battery Testing
- How to assemble and test a three electrode pouch cell? 2026 post
- Electrochemical Test Techniques for Lithium-ion Batteries: CV, EIS