How External Compressive Stress Influences Battery Performance?
Lithium-ion batteries represent the cornerstone of modern energy storage technology, and optimizing their performance remains a primary focus of current research. In practical applications, battery cells are frequently subjected to mechanical stress arising from module packaging constraints and volumetric changes induced by internal ionic diffusion. How this external compressive stress influences electrochemical behavior—specifically the underlying mechanisms in cathode materials like Lithium Iron Phosphate (LiFePO4)—remains a subject of ongoing investigation.
While LiFePO4 is widely utilized for its high safety profile and long cycle life, its charge-discharge process involves a two-phase transition that results in a flat voltage plateau, posing significant challenges for state-of-charge (SoC) estimation. Understanding the impact of stress on the electromechanical coupling of LiFePO4 is critical for the development of high-performance electrode materials and advanced battery designs.
This study reveals key findings through a combination of experimental analysis and theoretical modeling. The research team demonstrates that within a range of 0–0.9 MPa, external compressive stress can enhance the ionic diffusion coefficient of the LiFePO4 cathode, improve nucleation kinetics, and boost reaction reversibility. However, it was also observed that such stress slightly reduces the electrode’s equilibrium potential. These insights provide a novel framework for the stress-optimized design of high-efficiency battery systems.
Paper Title: Mechanistic Analysis on Electrochemo-mechanics Behaviors of Lithium Iron Phosphate Cathodes
Journal: Acta Materialia (IF=9.3)
DOI Link: https://doi.org/10.1016/j.actamat.2025.121024
Research Team: Huacui Wang, Binghe Liu, Dongjiang Li, Jun Xu (Corresponding Authors: Binghe Liu, Jun Xu)
1. Graphical Abstract and Introduction
This study systematically evaluates the electrochemical-mechanical coupling behavior of $LiFePO_4$ cathodes by applying controlled external mechanical stress. The research team developed a specialized three-electrode battery cell configuration to isolate cathode performance.
To uncover the underlying mechanisms of stress-induced changes in diffusion, impedance, nucleation, and equilibrium potential, the team employed a comprehensive suite of electrochemical and structural characterization techniques:
Galvanostatic Charge-Discharge (GCD): To evaluate capacity and voltage profiles.
Cyclic Voltammetry (CV): To study redox kinetics and reversibility.
Electrochemical Impedance Spectroscopy (EIS): To measure internal resistance and charge-transfer kinetics.
Galvanostatic Intermittent Titration Technique (GITT): To quantify the ionic diffusion coefficient.
In-situ X-ray Diffraction (XRD): To observe crystal lattice changes and phase transitions in real-time under varying stress levels.
Experimental Design and Material Characterization
The research was conducted using high-quality materials harvested from a commercial 86 Ah $LiFePO_4$ battery. After careful disassembly, the cathode material underwent rigorous baseline characterization:
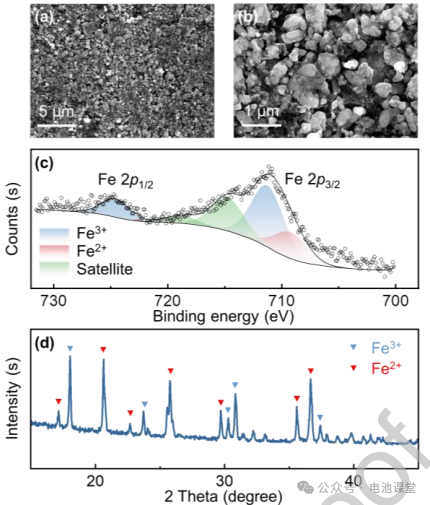
Surface Morphology: Scanning Electron Microscopy (SEM) imaging revealed a highly uniform granular structure of the $LiFePO_4$ electrode (Fig. 1a-b), which is critical for consistent stress distribution.
Chemical and Structural Composition: X-ray Photoelectron Spectroscopy (XPS) and XRD confirmed the precise chemical stoichiometry and the high crystalline quality of the olivine structure (Fig. 1c-d).
These baseline results established a robust foundation for the subsequent analysis of how external compressive stress modifies the electrochemical thermodynamics and kinetics of the battery system.
Figure caption: Material characterization shows that the LiFePO4 electrode particles are uniformly distributed and have a well-defined crystal structure, ensuring the reliability of the experimental samples.
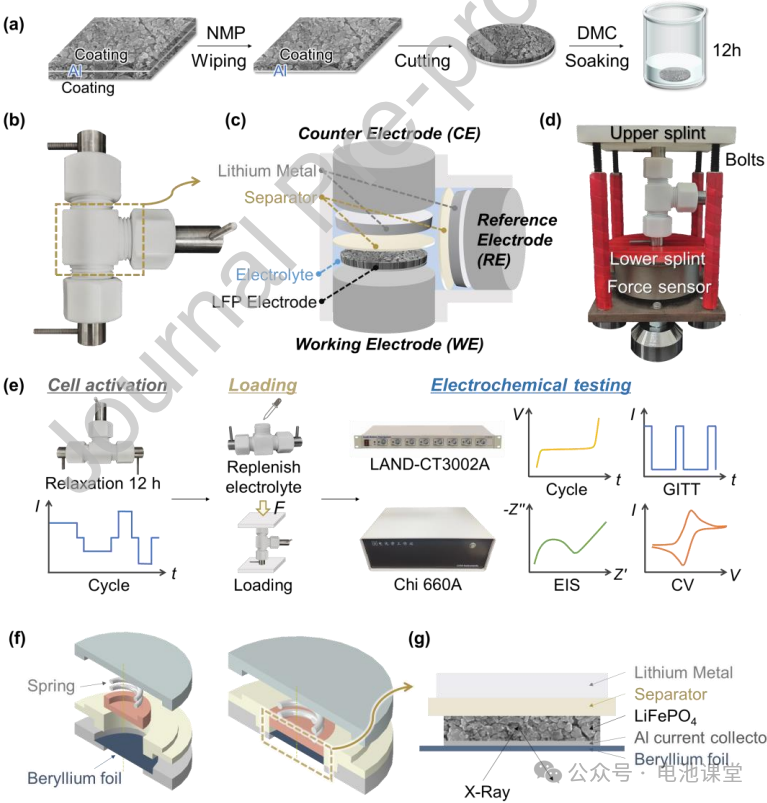
Stress application and battery assembly: A Swagelock-type three-electrode battery (Figures 2b-c) was used, and compressive stresses of 0, 0.3, 0.6, and 0.9 MPa were applied using a custom fixture (Figure 2d). Battery assembly was performed in an argon-filled glove box, using lithium metal as both the counter and reference electrodes to ensure testing accuracy. The experimental procedures included battery activation, stress loading, and electrochemical testing (Figure 2e), avoiding the influence of sample differences on the results.
Figure caption: The experimental design ensured the uniformity and repeatability of stress application, providing a reliable platform for subsequent electrochemical behavior analysis.
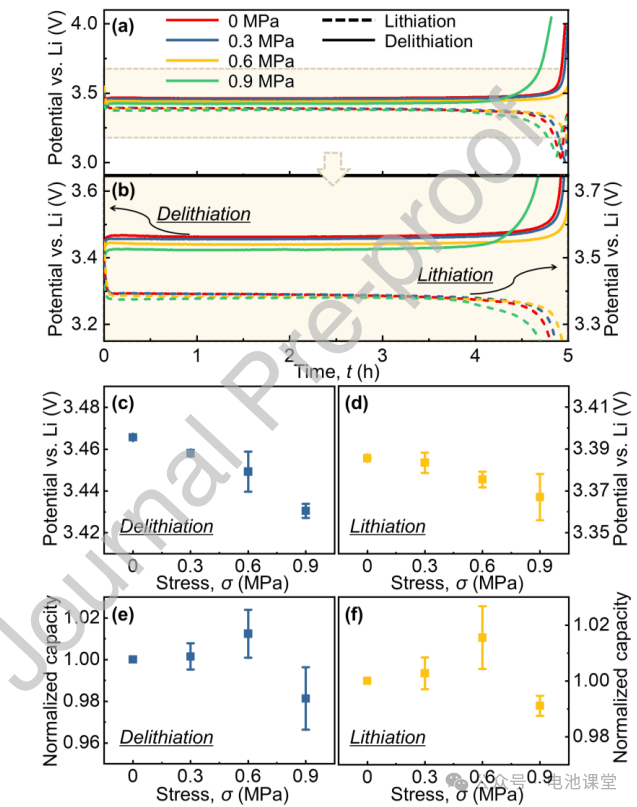
Effect of stress on charge-discharge behavior: Constant current charge-discharge tests showed that the charge-discharge voltage plateau of LiFePO4 decreased significantly with increasing stress (Figures 3a-d). At 0.9 MPa stress, the voltage plateau for the delithiation process decreased by approximately 40 mV, while the decrease was smaller for the lithiation process. The capacity change showed a trend of first increasing and then decreasing, with the highest capacity at 0.6 MPa (Figures 3e-f), indicating that moderate stress can improve performance, but excessive stress may induce side reactions.
Figure caption: Stress affects voltage plateau and capacity by altering electrode reaction kinetics, highlighting the electrochemical-mechanical coupling effect.
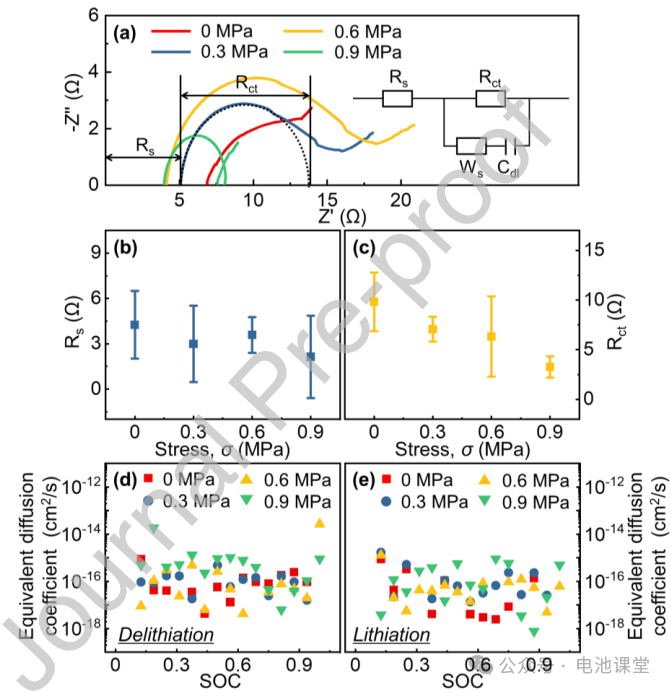
Enhancing effect of stress on impedance and diffusion: EIS tests show that both ohmic impedance (Rs) and charge transfer impedance (Rct) decrease with increasing stress (Figures 4a-c), while GITT calculations show an increase in the equivalent diffusion coefficient (Figures 4d-e). This indicates that compressive stress promotes ion migration and interfacial reactions, but the impedance change is not the primary cause of the voltage plateau decrease.
Figure caption: Stress optimizes the kinetic performance of the electrode by reducing impedance and increasing diffusion coefficient.
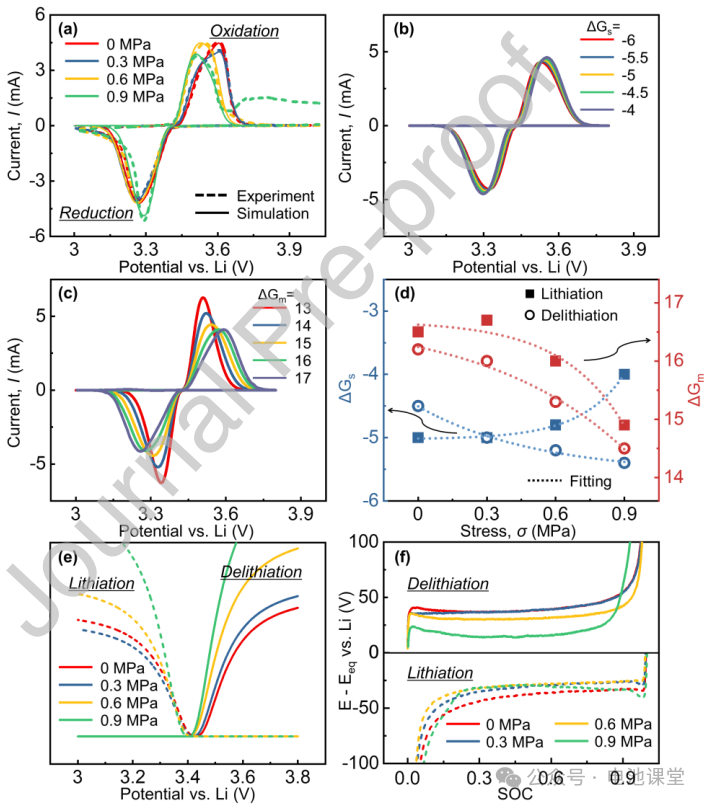
Stress regulation of nucleation: CV tests show that increased stress shifts the oxidation peak to the left, increases the reduction peak current, and improves reaction reversibility (Figure 5a). Based on the slow nucleation model and Gibbs free energy theory, simulations show that stress reduces the activation energy for atom migration (ΔGm), but decreases the mismatch strain energy (ΔGs) for delithiation, while increasing ΔGs for lithiation intercalation (Figures 5d-e). This leads to an overall increase in nucleation rate and a decrease in overpotential (Figure 5f), explaining the change in voltage plateau.
Figure caption: Stress enhances reactivity and uniformity by adjusting the nucleation energy barrier, providing theoretical support for the phase transition mechanism.
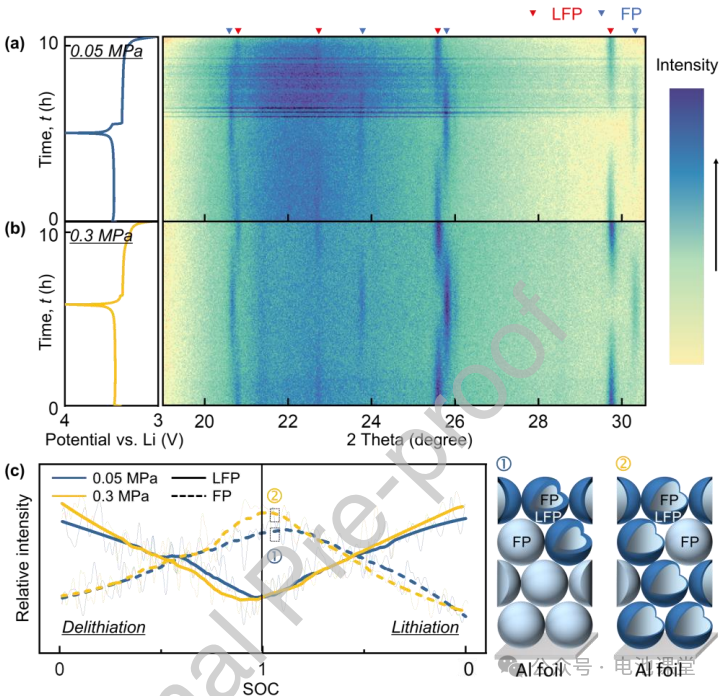
In-situ XRD verification of stress-driven phase transition uniformity: In-situ XRD experiments (Figures 6a-b) show that under high-pressure stress, the two-phase transitions of LiFePO4 and FePO4 are more synchronized (Figure 6c), indicating that stress increases the nucleation rate and makes the phase transition more uniformly distributed in the electrode particles.
Figure caption: Stress promotes a uniform phase transition in electrode particles, reduces reaction hysteresis, and verifies the positive influence of mechanical action on electrochemical behavior.
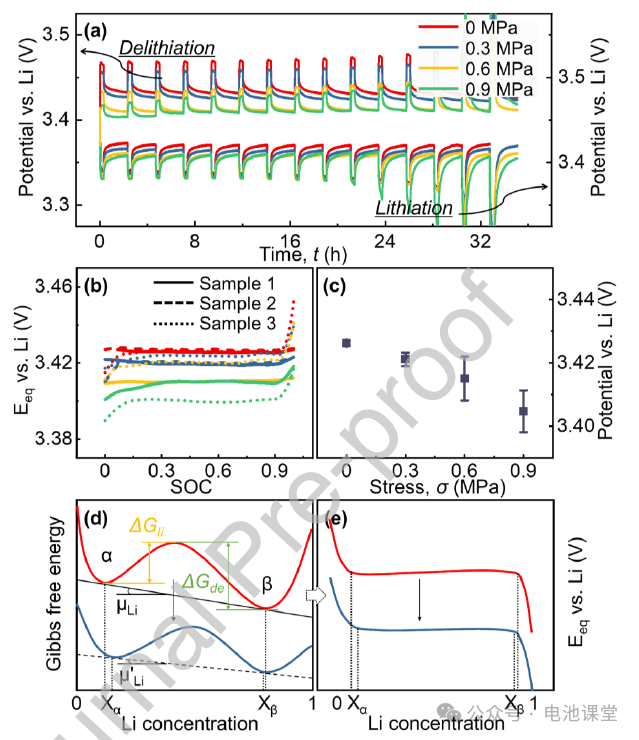
Effect of stress on equilibrium potential: GITT tests show that increased stress decreases the equilibrium potential (Figures 7a-c). Gibbs phase rule analysis shows that stress reduces the electrode’s equilibrium potential by altering the slope of the common tangent in the two-phase regions (Figures 7d-e), consistent with the decrease in the charge-discharge plateau.
Caption: Stress regulates the equilibrium potential through thermodynamic mechanisms, providing a complete explanation for electrochemical-mechanical coupling.
2. Summary and Outlook
Summary: This study confirms that external compressive stress significantly modulates the electrochemical-mechanical coupling of LiFePO4 cathodes. By integrating experimental data with theoretical modeling, the research demonstrates that applying external compressive stress within the 0–0.9 MPa range effectively enhances the ionic diffusion coefficient, optimizes nucleation kinetics, and improves reaction reversibility. These findings provide a fundamental explanation for the observed changes in equilibrium potential and impedance under mechanical constraints.
Outlook: Future research should focus on optimizing the uniformity of stress distribution across the electrode surface and investigating the effects of stress on other critical components, such as separators and anodes. These insights offer valuable guidance for the structural design of battery modules. By implementing precise stress control strategies, it is possible to significantly enhance the overall power density, cycle life, and operational stability of LiFePO4 battery systems.
3. About the Authors
Corresponding Author: Jun Xu
Professor in the Department of Mechanical Engineering at the University of Delaware and Director of the Mechanics of Energy & Sustainability Laboratory. His research focuses on multiphysics coupling modeling and the mechanical behavior of battery materials. He has published numerous high-impact papers in the interdisciplinary field of electrochemistry and mechanics.
Corresponding Author: Binghe Liu
Researcher at the School of Mechanical and Vehicle Engineering, Chongqing University. His expertise lies in battery safety and the optimization of mechanical performance in energy storage systems.
First Author: Huacui Wang
PhD candidate at Chongqing University. Her research primarily involves experimental analysis and numerical simulation of lithium-ion battery electrode materials.
The collaborative efforts of this team have significantly advanced the mechanistic exploration of mechanical behavior in advanced batteries.
Mechanistic analysis on electrochemo-mechanics behaviors of lithium iron phosphate cathodes
Originality Statement
This article was interpreted and authored by “Battery Classroom” based on publicly available academic papers. We welcome professional sharing and exchange. Please cite the source for any reprints. For copyright concerns or removal requests, please contact us for immediate rectification.
Pressure Fixture / Compression Fixture / Compression Plates for pouch cells

Due to the limited knowledge and English level is inevitable errors and omissions, if there are errors or infringement of the text, please contact me as soon as possible by private letter, I will immediately be corrected or deleted.