Breaking the Fast-Charging Ceiling!
Mingjian Zhang (CUHK), Zuwei Yin (XMU) & Zhiman Liu (Li Auto) in Matter: Ultrafast Sintering Post-treatment Unlocks High-Performance olivine type cathodes

First Authors: Yuansheng Lin, Enze Li
Corresponding Authors: Mingjian Zhang, Zuwei Yin, Zhiman Liu
Equipment Used: This study utilized the Neware MIHW-200-160CH All-in-one Integrated Coin Cell Testing System.
Research Background
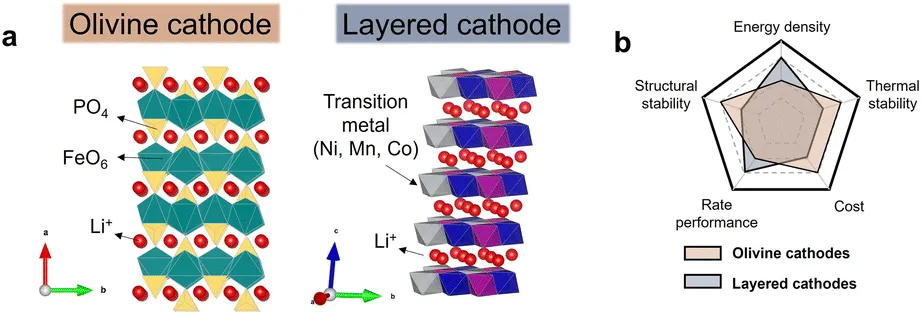
In the wave of next-generation fast-charging technology, a vast amount of existing commercial olivine-type cathode materials (such as lithium iron phosphate, LFP) are facing severe performance bottlenecks. Directly discarding or blindly recycling them would cause immense resource waste and environmental pressure.
An intuitive approach to enhancing the fast-charging capability of LFP is to increase the crystallinity of the surface carbon layer to improve electronic conductivity. However, traditional carbothermal synthesis is limited by relatively low temperatures (600–800°C), which only allows for the formation of amorphous carbon layers with poor conductivity. If the sintering temperature is blindly increased, according to Arrhenius kinetics, the grain size will undergo significant coarsening, which severely deteriorates the rate performance of the material.
How can we break this “you can’t have your cake and eat it too” paradox? Specifically, achieving a substantial increase in carbon layer crystallinity without causing nanoparticle coarsening has become the core pain point in breaking through the fast-charging ceiling of commercialized olivine materials.
Research Highlights
Recently, the research group led by Mingjian Zhang from The Chinese University of Hong Kong, Shenzhen, in collaboration with Zuwei Yin from Xiamen University and Zhiman Liu’s team from Li Auto, developed a simple and scalable ultrafast sintering strategy to address this issue.
Inspired by Zener pinning-constrained grain growth kinetics, this strategy eliminates the need for complex doping or coating treatments. Instead, it involves exposing synthesized commercial cathode materials to a brief sintering process (rapidly heating to 1000°C in just over 10 seconds, followed by immediate cooling).
This treatment simultaneously optimizes the surface carbon layer (reducing C-O defects) and the bulk microstructure (doubling the amount of Fe/Li antisite defects) of the LFP material, all while maintaining the original nanometer-scale grain size. As a result, both the surface electronic conductivity and bulk lithium-ion diffusion capabilities were significantly enhanced.
The upgraded LFP and Li(FeₓMn₁₋ₓ)PO₄ cathodes demonstrated a rate capability improvement of over 25% and a significantly extended cycle life (with only 9.8% capacity decay after 5,000 fast-charging cycles). Furthermore, this approach shows broad applicability for future high-energy-density solid-state lithium metal batteries.
Crucially, by simply retrofitting a standard tube furnace, this method has already achieved kilogram-scale engineering scale-up in the laboratory. This research provides a transformative methodological innovation for improving the performance of commercial materials—prioritizing upgraded processing over re-synthesis or recycling—paving the way for the industrial production of next-generation high-performance cathodes.
This work was published in Matter (the flagship materials science journal under Cell Press). The first authors of the article are PhD student Yuansheng Lin and Master’s student Enze Li from The Chinese University of Hong Kong, Shenzhen.
Main contents of the research
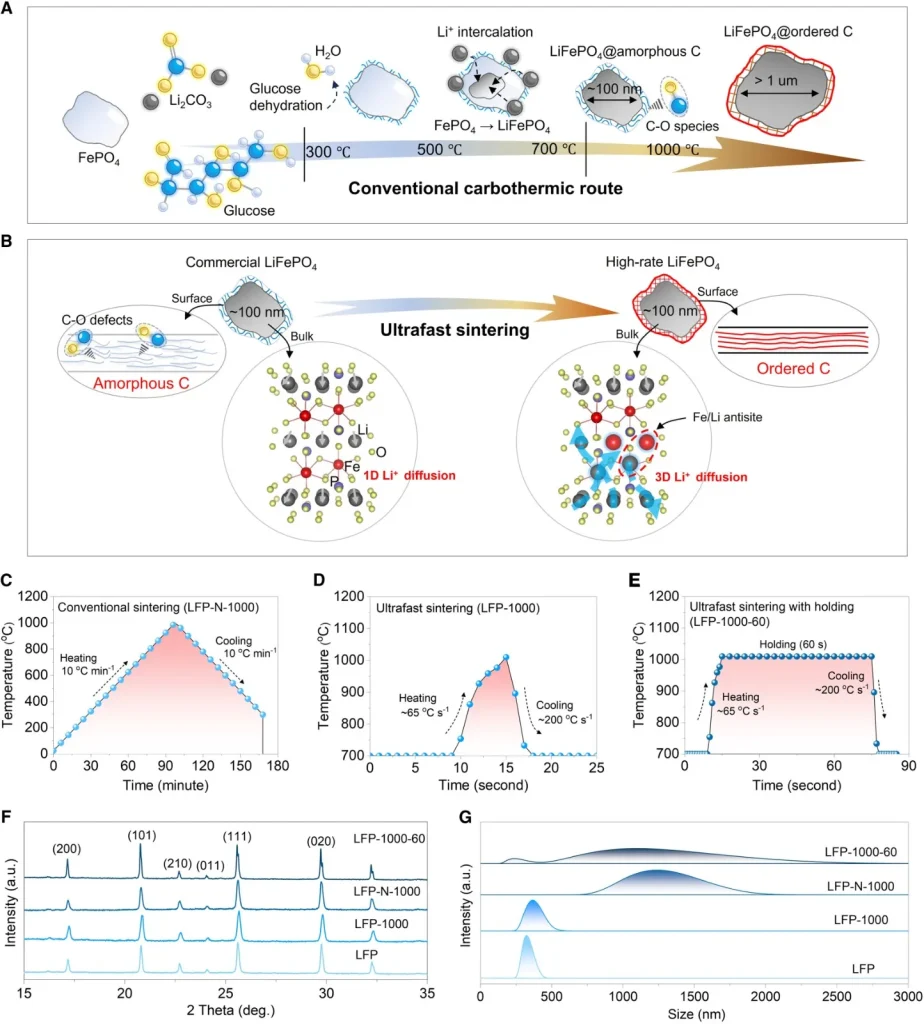
1. Breaking Traditional Limits: Simultaneous Modulation of Surface and Bulk Phases
The study reveals that traditional sintering modes—characterized by slow heating and prolonged holding times—inevitably lead to the growth of LFP primary particles into the micrometer scale, which severely degrades electrochemical performance. Through systematic exploration, the team identified an optimal ultrafast sintering process window: a heating rate of 65°C/s, a peak temperature of 1000°C, and a cooling rate of 200°C/s.
This non-equilibrium thermal treatment perfectly bypasses the particle coarsening zone. Simultaneously, it drives the evolution of the surface carbon layer toward an ordered structure and drastically reduces C-O defects, leading to a leap in both powder conductivity and charge-transfer kinetics.
Multi-dimensional characterization confirms that after ultrafast sintering, the thickness of the LFP surface carbon layer is reduced, its structural order is significantly enhanced (indicated by a decrease in the Raman ID/IG ratio), and the lattice fringes appear much clearer compared to untreated samples. Electron Energy Loss Spectroscopy (EELS) results show that the surface of untreated commercial LFP samples contains significant broadening characteristic peaks corresponding to C-O defects within the first few atomic layers. In contrast, the modified LFP-1000 sample shows no obvious C-O defects at the same depth and exhibits sharp characteristic peaks corresponding to lattice oxygen.
Powder compaction conductivity tests demonstrate that the electronic conductivity of LFP is significantly improved after ultrafast sintering. Furthermore, kinetic analysis based on Distribution of Relaxation Times (DRT) spectra confirms that LFP-1000 exhibits charge-transfer kinetics far superior to those of untreated commercial samples during high-rate charging and discharging.
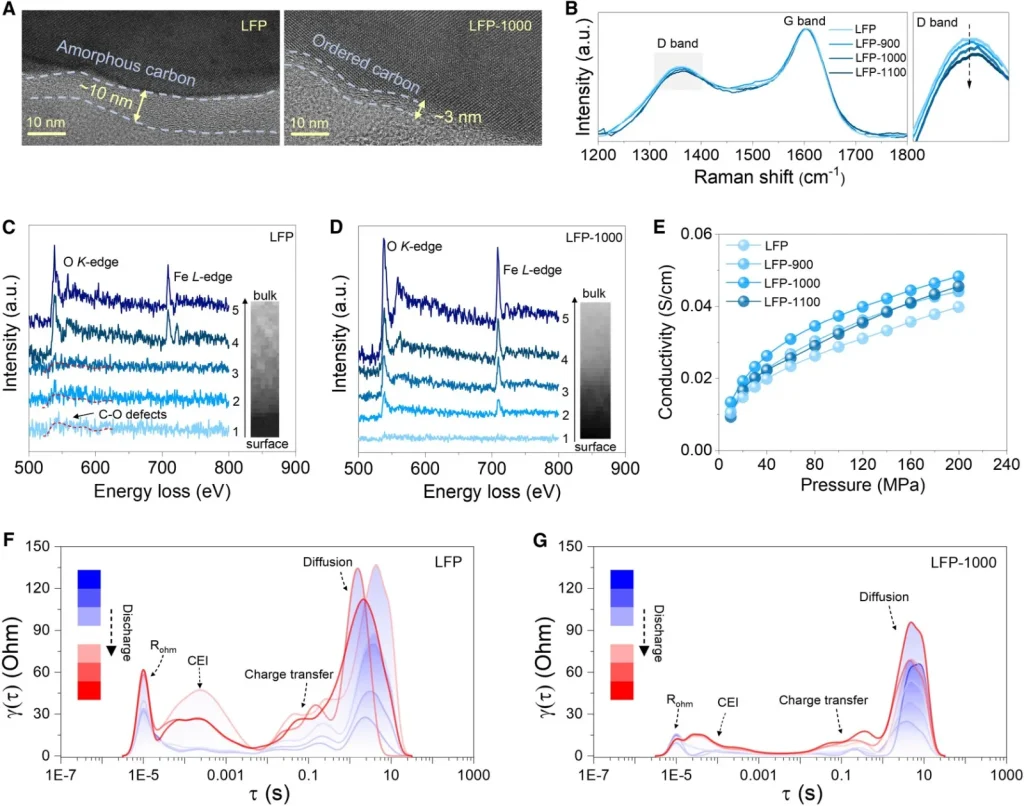
(B) Raman spectra of LFP samples at different sintering temperatures.
EELS spectra measured along the direction from the surface to the bulk for (C) LFP particles and (D) LFP-1000 particles.
(E) Curves of powder electrical conductivity versus pressure for LFP, LFP-900, LFP-1000, and LFP-1100.
DRT analysis results at 8C for (F) LFP and (G) LFP-1000 half-cells.
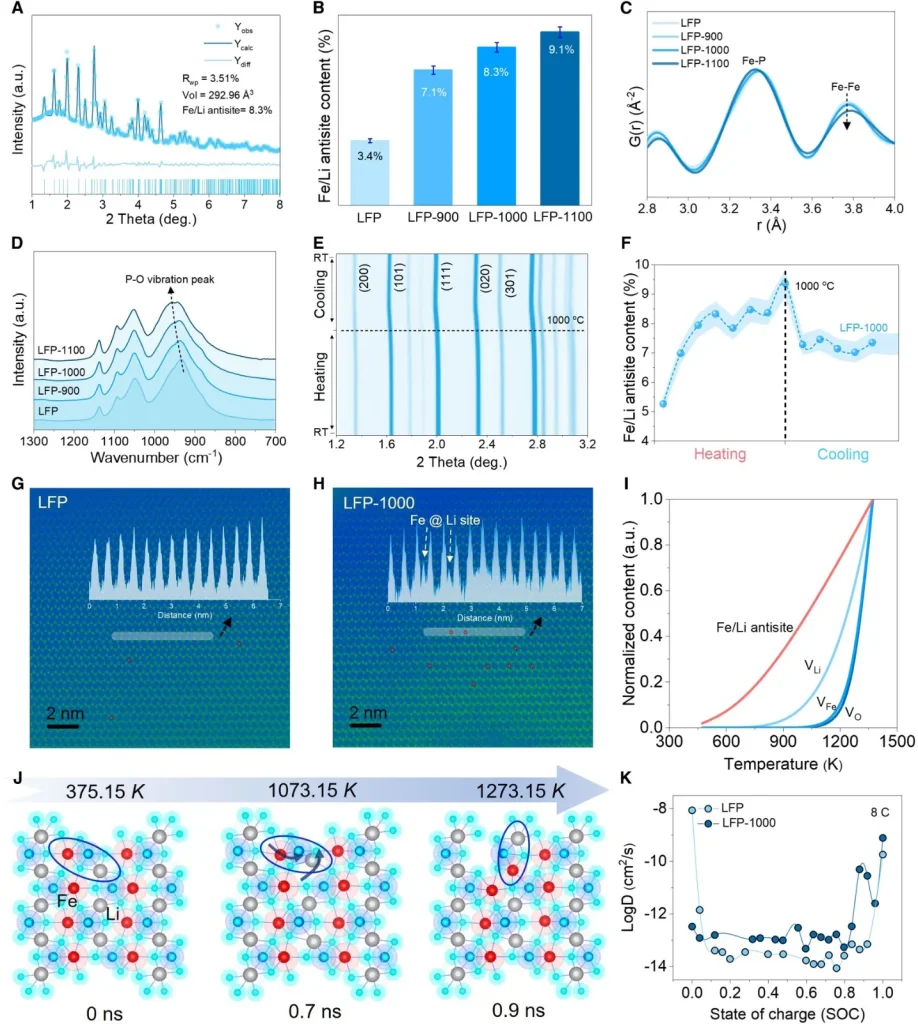
2. Exploring Bulk Microstructures: Thermally Driven Antisite Defects and Kinetic Enhancement
To uncover the microscopic mechanism by which ultrafast sintering boosts ion diffusion, the team combined synchrotron X-ray total scattering (PDF), in-situ SXRD, FTIR, and ab initio molecular dynamics (AIMD) simulations. The study confirms that the rapid high-temperature shock “freezes” a higher concentration of Fe/Li antisite defects within the LFP bulk. As temperature rises, the concentrations of oxygen, iron, and lithium vacancies in the lattice increase sharply. At approximately 1073.15 K, thermally driven site exchange occurs between Fe and Li atoms, thereby opening up three-dimensional (3D) lithium-ion diffusion pathways. This significant leap in the diffusion coefficient was further validated by GITT measurements.
3. Full-Scenario Performance Validation and Kilogram-Scale Mass Production
The processed olivine-structured cathode materials demonstrate remarkable electrochemical upgrades:
Liquid-State Fast Charging: The discharge capacity at 8C is improved by approximately 25% compared to the original commercial sample, with an impressive capacity retention of 90.2% after 5,000 cycles.
Solid-State Batteries: In both 60°C all-solid-state and 25°C semi-solid-state batteries, its high-rate capacity significantly outperforms (crushes) the original commercial counterparts with an absolute advantage.
Universality and Mass Production: This technology not only revitalizes commercial lithium manganese iron phosphate (LMFP)—doubling its 8C capacity and boosting the 1,000-cycle retention rate from 34.9% to 69.6%—but has also achieved kilogram-scale continuous preparation through an automated conveyor system operating at 18 cm/s. Theoretically, ton-scale mass production can be realized with only minor modifications to industrial roller hearth kilns.
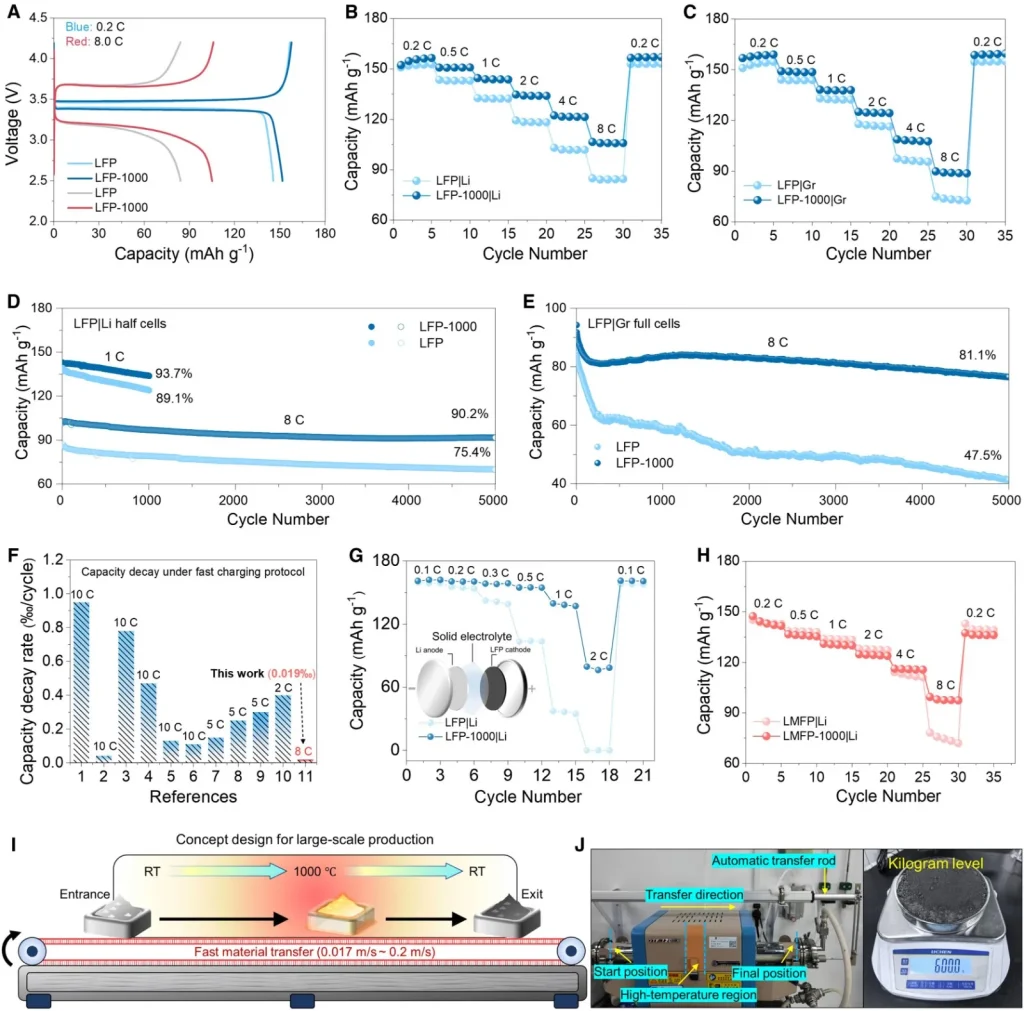
4. Unveiling the Mechanism for Ultra-Long 8C Cycle Life (5,000 Cycles): Carbon Layer Optimization Inducing Deep Remodeling of Interfacial Chemistry
As illustrated in Figure 5, to explore the underlying mechanism for the enhanced cycling stability after ultrafast sintering, structural and compositional analyses were performed on the cathodes after 5,000 cycles at 8C using TOF-SIMS, XPS, XRD, SEM, and HRTEM. As previously discussed, XPS and EELS results indicate that ultrafast sintering induces a reduction of C-O species on the surface carbon layer, which suppresses the oxidative decomposition of organic solvents at high potentials.
Prior research has shown that ethylene carbonate (EC) undergoes multi-step oxidation at the electrode/electrolyte interface to generate H2O; this, in turn, accelerates the hydrolysis of carbonate solvents (such as EC and ethyl methyl carbonate), triggering continuous parasitic side reactions. Meanwhile, the generated H2O molecules exacerbate the hydrolysis of the LiPF6 electrolyte salt, producing a significant amount of hydrofluoric acid (HF), which attacks the crystal structure of the cathode material. Consequently, the surface of cycled LFP exhibits a higher content of organic components, and evident intragranular cracking was observed via HR-TEM. The suppression of C-O defects by ultrafast sintering essentially blocks the initial steps of these continuous side reactions, thereby achieving compositional optimization of the CEI (Cathode Electrolyte Interphase) and significantly improved cycling stability.
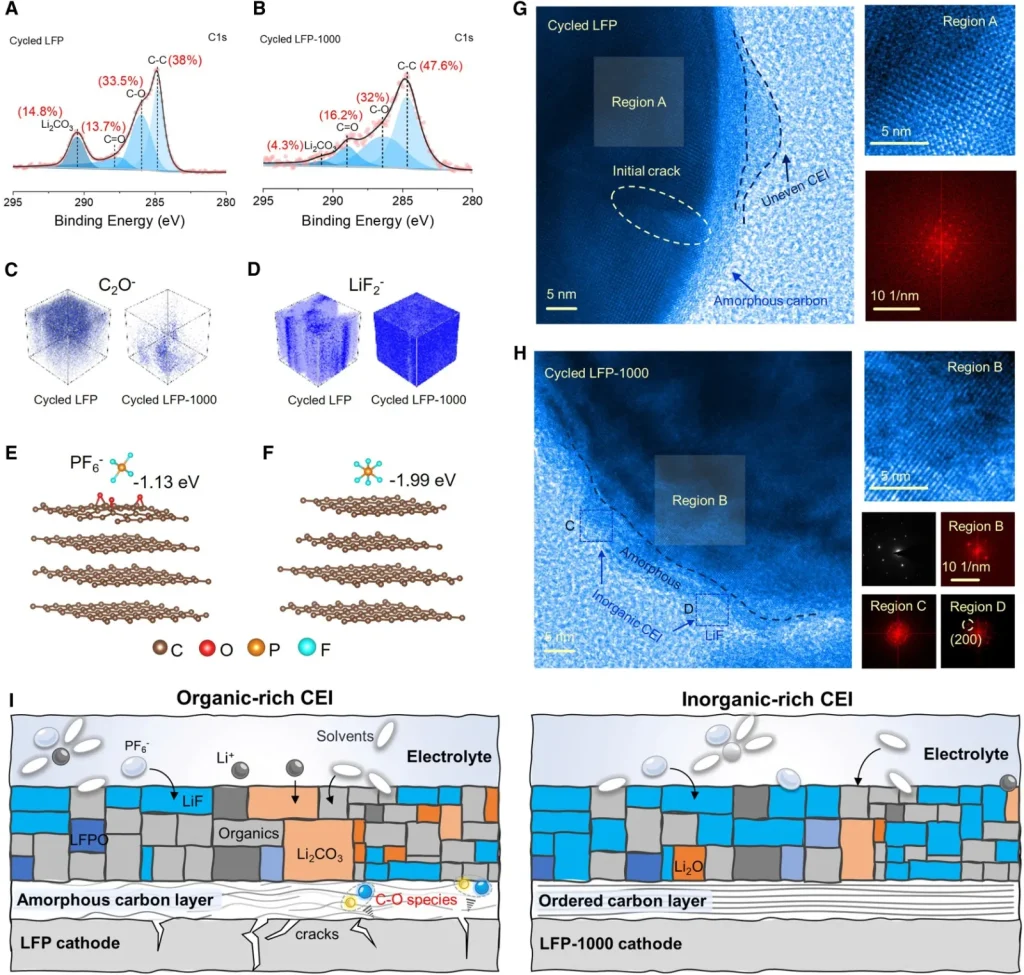
(C, D) 3D mapping of (C) C2O- and (D) LiF2- fragments from TOF-SIMS results.
DFT simulations of PF6- anion adsorption behavior on carbon layers (E) with C-O defects and (F) without C-O defects.
HRTEM images of (G) LFP and (H) LFP-1000 after 5,000 cycles at 8C.
(I) Schematic illustration (cartoon model) of the CEI layers in LFP and LFP-1000 after long-term cycling.
Conclusion and Outlook
This study transcends the traditional constraints of “novel material synthesis” by ingeniously applying grain growth kinetic theory to pioneer an “ultrafast sintering upgrade” technology for commercial olivine-structured cathodes. By achieving precise, synchronous reconstruction of the “surface carbon layer” and “bulk defects” at the nanoscale, this approach effectively resolves the long-standing trade-off between fast-charging performance and material stability. This work not only provides a highly cost-effective shortcut for the industrial upgrading of sustainable lithium-ion batteries but also offers valuable insights for breaking performance bottlenecks in other carbon-coated systems, such as silicon-carbon (Si/C) and graphite materials.
Equipment Details
The electrochemical tests in this study were conducted using a NEWARE All-in-one for 160 coin cells (Model: MIHW-200-160CH). For inquiries or procurement, please contact the manufacturer through the following channels: Neware all in one


More details: Simultaneous regulation of surface-bulk microstructure to achieve long-term fast-charge olivine-type cathodes