Accelerating Anode-free Lithium Metal Battery R&D with Neware’s High-Precision Battery Testing Solutions

High-Energy Density Anode-Free Lithium Metal Batteries (AFLMBs) via Cross-Coupled Interfacial Chemistry
Digital Object Identifier (DOI): https://doi.org/10.1038/s41586-026-10402-0
Affiliation: Westlake University
Nature Published: 17 March 2026
Abstract Overview
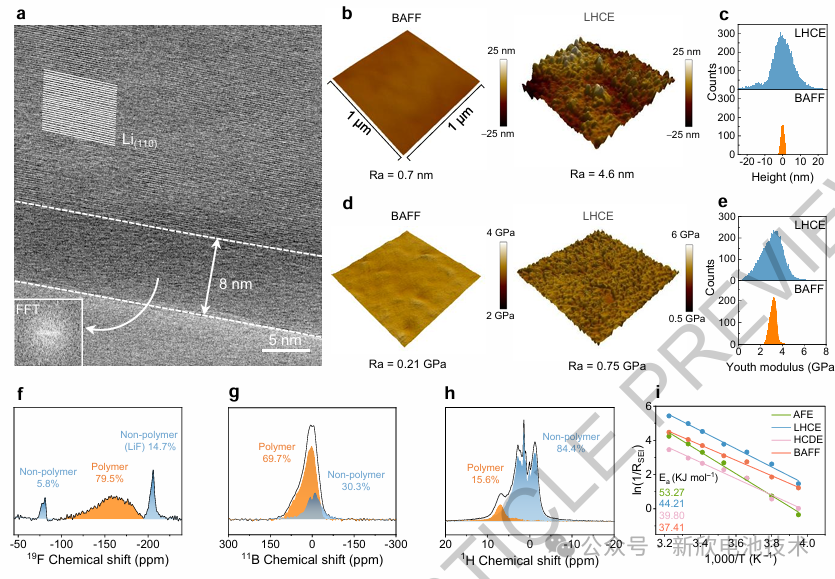
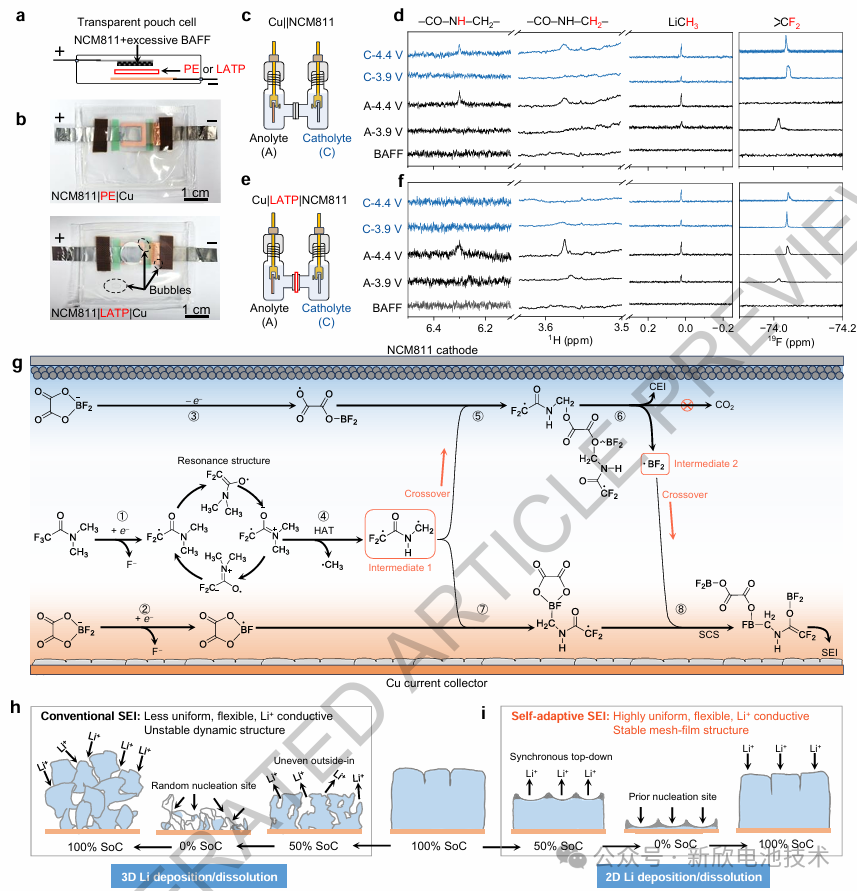
Anode-free lithium metal batteries (AFLMBs) eliminate the need for initial negative active materials during assembly, presenting a transformative pathway toward ultra-high energy density and cost-efficient energy storage. However, the inherent absence of lithium reservoirs and host frameworks imposes rigorous operational constraints, typically resulting in compromised cycle life. These longevity issues are fundamentally linked to inhomogeneous lithium plating/stripping, driven by the microscopic heterogeneity and mechanical brittleness of the Solid Electrolyte Interphase (SEI).
This research introduces a practical 500 Wh kg⁻¹-class AFLMB utilizing a novel cross-coupled electrolyte system. The electrolyte initiates synergistic interfacial reactions that cultivate a B-F polymer-rich SEI at the anode while simultaneously suppressing parasitic gas evolution at the cathode. The resultant interphase layer exhibits sub-nanoscale homogeneity, exceptional mechanical flexibility, and accelerated Li+ transport kinetics. Furthermore, it spontaneously forms an adaptive network-like membrane that harmonizes ionic flux and accommodates significant volumetric fluctuations.
Performance Benchmarks
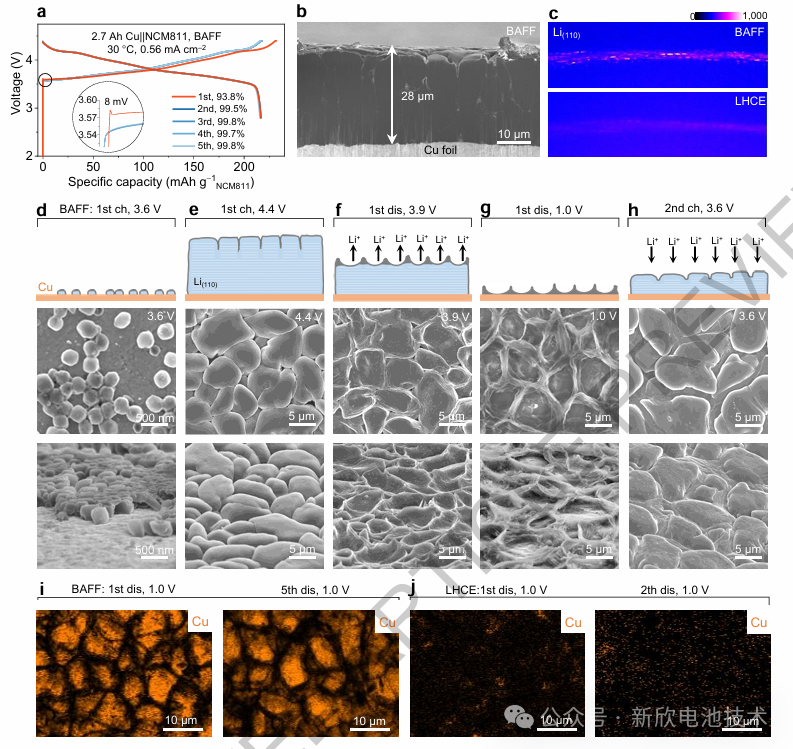
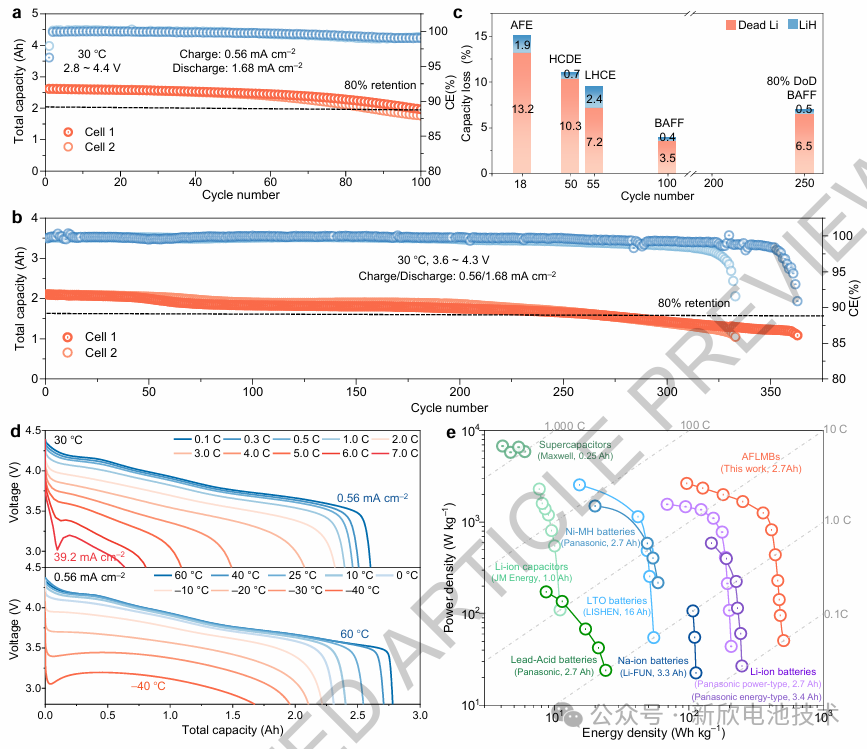
The implementation of this interfacial chemistry enables a reversible planar lithium plating/stripping capacity of 5.6 mAh cm⁻². Consequently, a host-less 2.7 Ah AFLMB (achieving 508 Wh kg⁻¹ and 1668 Wh L⁻¹) demonstrated robust stability:
100% Depth of Discharge (DoD): Sustained over 100 cycles.
80% Depth of Discharge (DoD): Maintained 80% capacity retention over 250 cycles.
Power Capability: Achieved a high power density of 2650 W kg⁻¹ at an energy density of 96 Wh kg⁻¹.
These findings validate the efficacy of cross-coupled interfacial engineering in mitigating the structural instabilities inherent to host-less electrodes, marking a significant milestone in the commercial viability of AFLMB technology.
Experimental Section: Electrolyte Synthesis and Physico-chemical Characterization
Reagent Procurement and Purification
High-purity electrolyte components, including LiFSI, LiPF₆, LiDFOB, and LiBF₄ (all 99.9% grade), alongside solvents such as DEC (99.99%), FEC (99.9%), FEMC (99.8%), and DME (99.9%), were sourced from Duoduo Chemical Co. Commercial carbonate electrolytes (1.0 M LiPF6 in EC/DMC) were utilized as benchmarks. Diluents and specialized additives, including HFE (>99.0%), TTE (>95%), and NDFA (>98%), were procured from TCI (Tokyo Chemical Industry Co., Ltd.). To ensure anhydrous conditions, all solvents were desiccated over 3 Å molecular sieves (Aladdin) for 48 hours prior to formulation.
Electrolyte Formulation Protocols
Four distinct electrolyte systems were synthesized with precise stoichiometric ratios:
AFE Electrolyte: 1.0 M LiPF6 dissolved in a ternary mixture of FEC/FEMC/HFE (2:6:2 by weight).
LHCE (Localized High-Concentration Electrolyte): 1.5 M LiFSI in a DME/TTE binary system (1.2:3 molar ratio).
HCDE (High-Concentration Dual-Salt Electrolyte): 2.0 M LiDFOB and 1.4 M LiBF4 in FEC/DEC (1:2 by volume).
BAFF Electrolyte: 1.6 M LiDFOB formulated in NDFA (1:5 molar ratio).
All synthesis procedures were executed within an argon-filled glovebox (Mikrouna; O2 < 0.1 ppm, H2O < 0.1 ppm) and maintained at ambient temperature.
Physico-chemical Analysis and Instrumentation
The moisture content of the resulting solutions was verified to be < 10 ppm via Karl Fischer titration. Technical characterization was conducted as follows:
Ionic Conductivity: Determined via Electrochemical Impedance Spectroscopy (EIS) using a Solartron 1470E workstation in a symmetric Pt|Electrolyte|Pt cell configuration.
Viscosity and Density: Measured using a kinematic viscometer (Anton Paar, SVM 3001).
Solvation Structure: Investigated through Raman Spectroscopy (Anton Paar, Cora 5700) employing a 785 nm excitation laser source.
Image and text excerpt


Accelerating Anode-free Lithium Metal Battery R&D with Neware’s High-Precision Battery Testing Solutions
Full article
Planar Li deposition and dissolution enable practical anode-free pouch cells