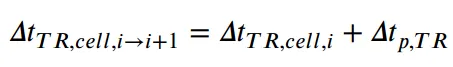
This paper presents a comparative study on the thermal runaway propagation of CATL batteries using NMC811 and LFP as cathode materials. The main conclusions are as follows:
Thermal Runaway Propagation Characteristics
The peak temperature during thermal runaway of NMC811 batteries (899°C) is significantly higher than that of LFP batteries (524°C). In addition, the thermal runaway propagation speed of NMC811 batteries is five times that of LFP batteries. This indicates that NMC811 batteries require additional safety measures, such as inter-cell spacing and thermal barriers, to meet safety standards.
Gas Emission
The gas volume, gas flow rate, and gas temperature released by NMC811 batteries during thermal runaway are all higher than those of LFP batteries. This requires the use of ventilation channels or high-temperature-resistant potting materials in NMC811 battery modules to isolate vented gases and avoid increased heat input, abrasion, and arcing risks.
Battery Damage
NMC811 modules exhibit higher risks of damage severity, material ejection, and side-wall rupture than LFP modules. Therefore, structurally reinforced potting materials or high-temperature-resistant materials (such as steel battery casings) are recommended to maintain the mechanical integrity of NMC811 batteries and ensure unobstructed vent gas discharge.
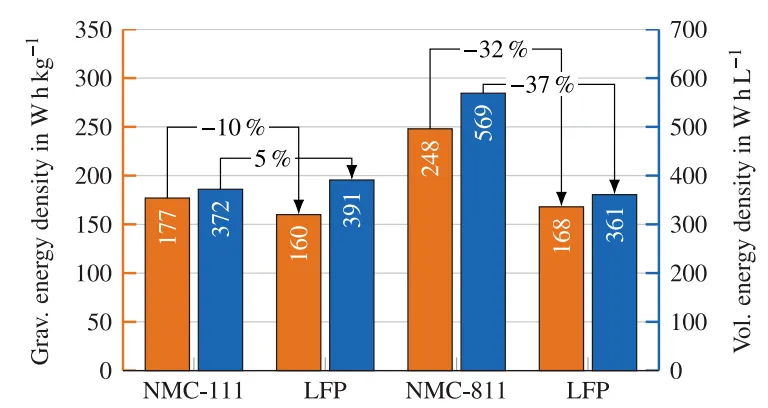
Due to their high nickel content, NMC811 batteries feature higher energy density. As shown in Figure 1, compared with LFP batteries, NMC811 batteries provide 32% higher gravimetric energy density and 37% higher volumetric energy density. In terms of safety, LFP batteries perform excellently and are less prone to overheating, combustion, and other hazardous conditions, benefiting from the stability and thermal resistance of their cathode materials.
To compare their thermal runaway propagation processes, CATL NMC811 and LFP batteries were selected for this study. Detailed specifications are listed in Table 1:
- NMC811 battery: 213 Wh/kg, 549 Wh/L, 96 Ah
- LFP battery: 164 Wh/kg, 373 Wh/L, 160 Ah
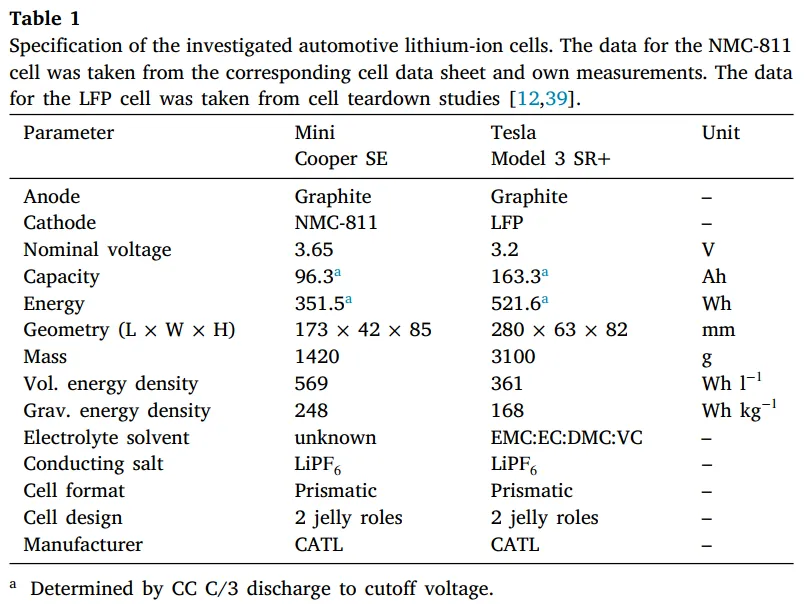
As shown in Figure 2, five batteries were assembled into a small module. A heating plate was installed on one side of the module to trigger thermal runaway by heating one of the batteries, with a heating power of 6.2 W/cm².
Thermocouples were placed between the batteries, on the battery surfaces, and near the safety valves to monitor temperature. The final experimental setup is shown in Figure 3, and a camera was used to record the thermal runaway process.
For accurate thermal runaway testing and precise temperature monitoring, Neware Battery Testing Systems paired with Neware Thermocouple Aux Channels are highly recommended.
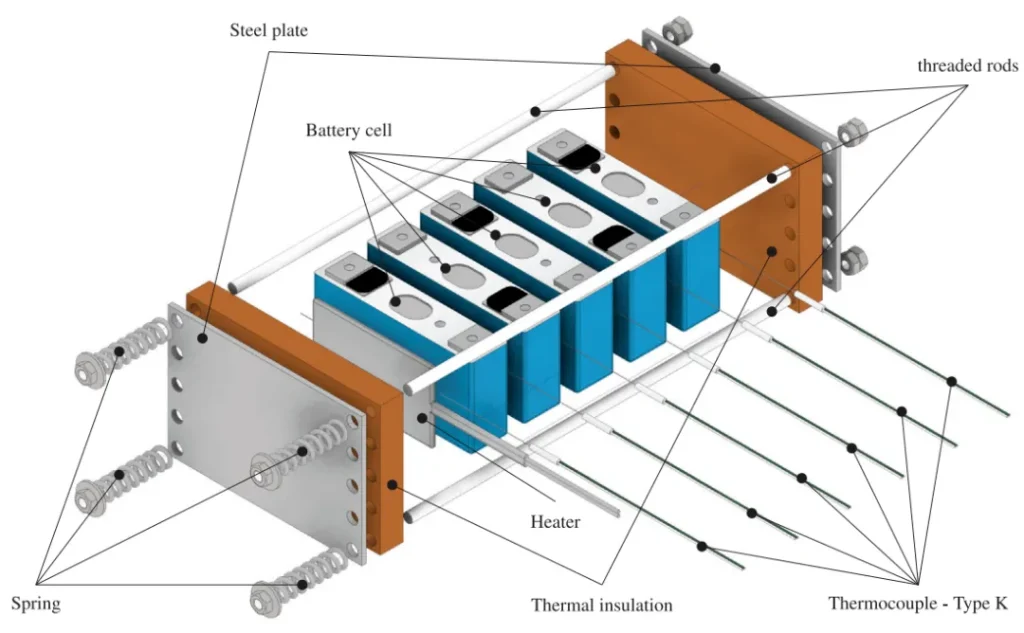

The start time of the heating plate heating stage is set as t0=0s. The onset of thermal runaway propagation is defined as the point when heating ends and temperature rises abruptly. The NMC811 module reached this point at a surface temperature of 371.6 °C. In contrast, thermal runaway in the LFP battery occurred much later, at 436.6 °C.
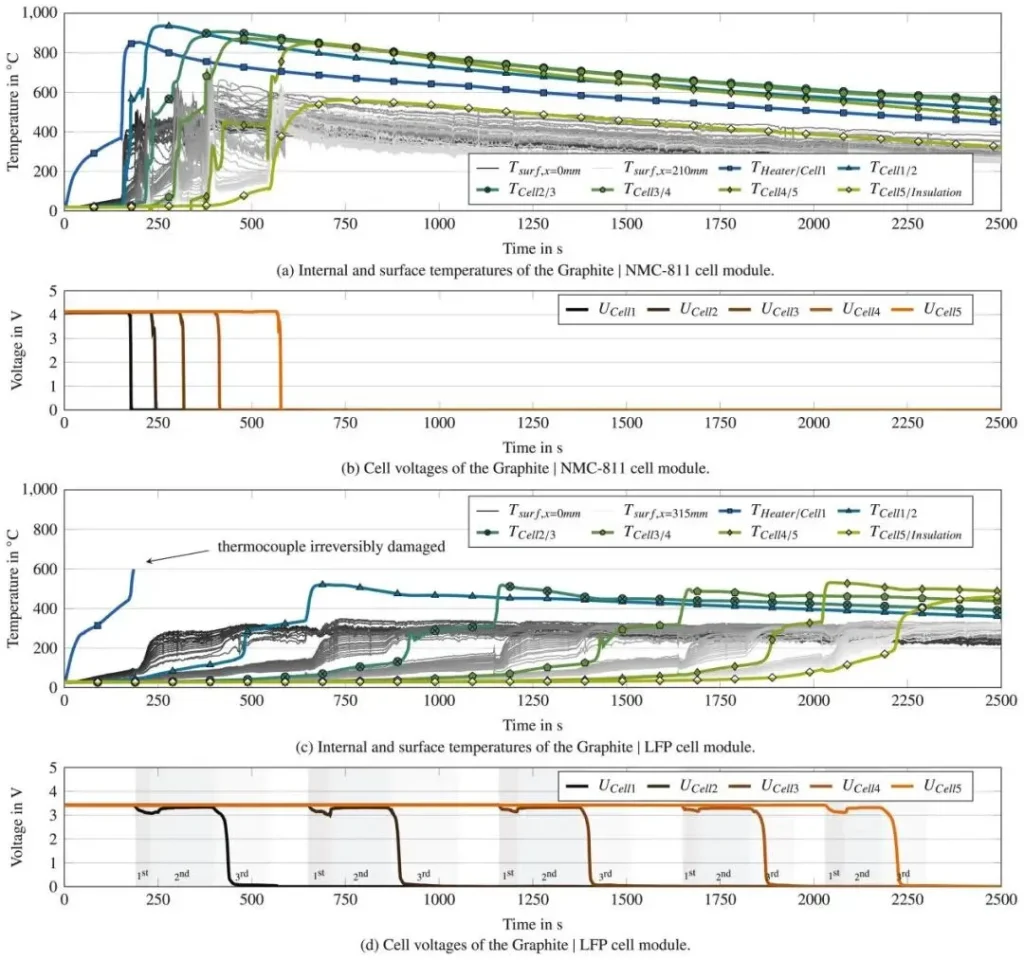
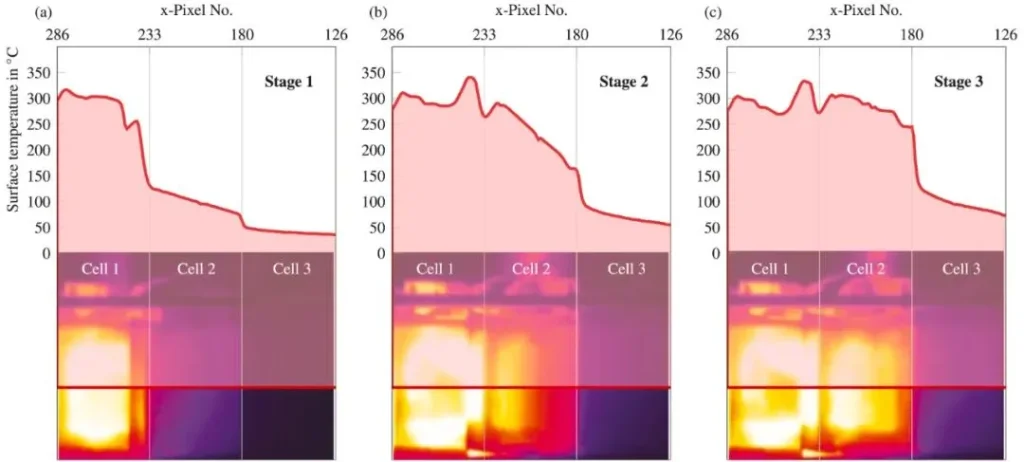
As shown in Figure 5, thermal runaway of a single cell (Cell 2 in Figure 5) within the battery module can be divided into three stages:
Stage 1: Heating caused by thermal runaway of the adjacent Cell 1;
Stage 2: Thermal runaway triggered in the first jellyroll of Cell 2;
Stage 3: Internal thermal runaway propagates to the second jellyroll in Cell 2.
The exact onset time of battery thermal runaway is determined by the following two criteria:
(1) The battery voltage begins to drop, i.e., the voltage change becomes negative;
(2) The temperature rise rate of any temperature sensor on the battery surface exceeds 10 K/s.
Based on the data shown in Figure 4 and the propagation mechanism illustrated in Figure 5, the time interval of thermal runaway between batteries can be further subdivided into the thermal runaway reaction time ΔtTR and the pause time ΔtP,TR. During the pause period, heat transfer to adjacent batteries mainly occurs.
For thermal imager and video data, the thermal runaway time is evaluated according to the visible time span of gas venting from the safety valve.
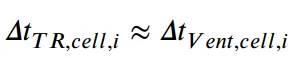
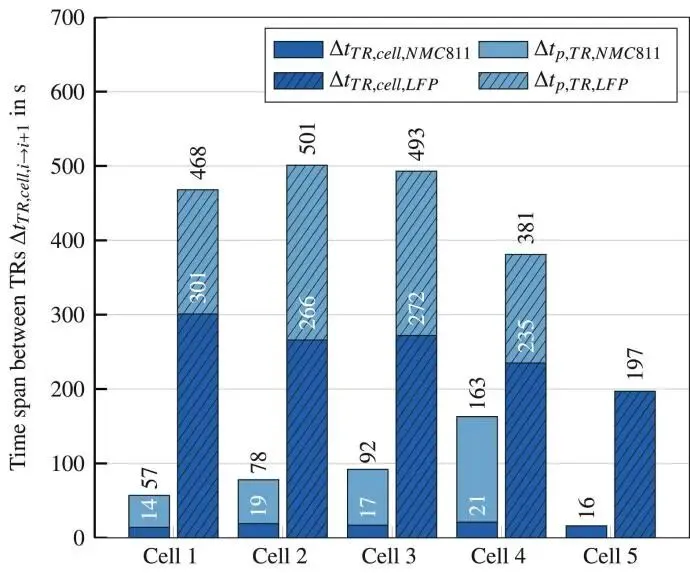
For LFP batteries, the time interval between thermal runaway events is distributed around an average value of 460 seconds, and the thermal runaway reaction time is distributed around an average value of 254.2 s.
For NMC811 batteries, the time interval between thermal runaway events increases exponentially, and the thermal runaway reaction time of NMC811 batteries is distributed around an average value of 17.4 s.
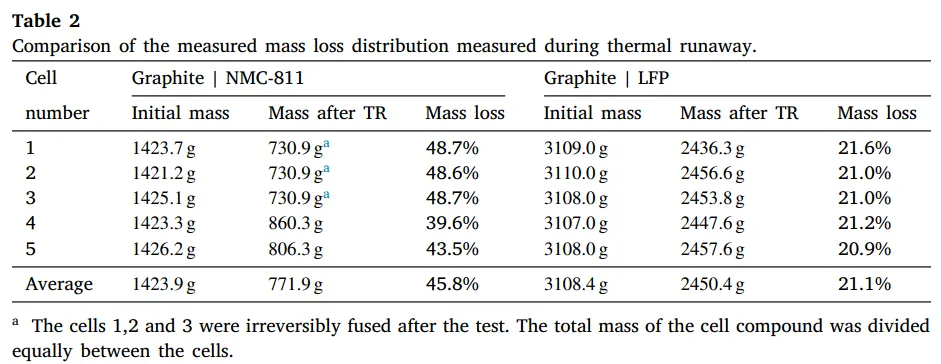

For more details, please refer to the reference:
Schöberl, J., Ank, M., Schreiber, M., Wassiliadis, N., & Lienkamp, M. (2024).
*eTransportation*, 19, 100305.
https://doi.org/10.14459/2023mp1717758