How to resolve the issue of CR2032 batteries being unable to support high-current discharge?
During the material R&D phase, it was observed that some CR2032 coin cells failed to discharge normally (Figure 1), which hindered the accurate and rapid acquisition of material performance data. This paper provides a fundamental analysis of why CR2032 lithium-ion coin cells fail to support high-current discharge. Through experimental troubleshooting, a definitive assembly strategy for CR2032 coin cells was established.
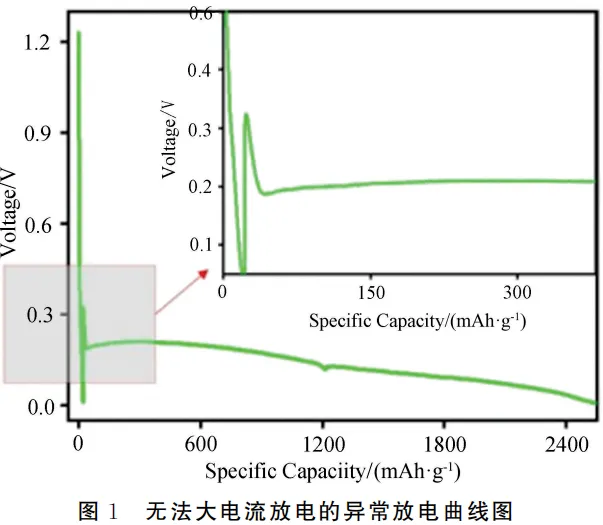
1. Introduction to the Assembly Process of CR2032 Lithium-ion Coin Cells
1.1 Instruments
Rolling machine (Calender); Punching machine; Coating machine; Glove box; Sealing machine; Battery testing system; Mixer.
1.2 Reagents
CR2032 coin cell cases; Copper foil (9μm); Binder (SBR, CMC); Conductive carbon black; Deionized water; Lithium metal chips; Separator; Electrolyte; Anode material (Silicon-carbon material).
1.3 Experimental Methods
The anode material, binder, and conductive carbon black (SP) were mixed in a mass ratio of 85:10:5 with an appropriate amount of deionized water to form a uniform slurry, which was then coated onto the copper foil. After drying at a specific temperature, the anode electrodes were obtained through rolling and cutting. The CR2032 coin cells were assembled using the anode electrode as the working electrode, lithium metal chips as the counter electrode, and a microporous polypropylene film as the separator, with a specific amount of electrolyte added.
The assembly of the CR2032 coin cells was conducted in an argon-filled glove box. The assembly schematic is shown in Figure 2. The assembly sequence followed: Positive case→ Spring (wave washer) → Spacer → Positive electrode (active material) → Separator → Lithium chip → Negative case. An appropriate amount of electrolyte was added to both sides of the separator before the battery was sealed. After completion, the CR2032 coin cells were left to stand for at least 12 hours.
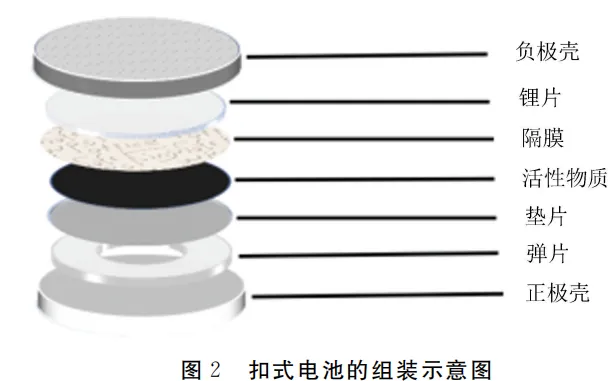
2. Analysis of the Causes for CR2032 Coin Cells’ Inability to Support Normal High-Current Discharge
In the research and development process of lithium-ion batteries, electrode thickness, sealing pressure, and baking conditions are three key factors that exert a crucial influence on the electrochemical performance of the battery.
(1) Electrode thickness has a significant impact on the internal resistance and electrochemical reaction kinetics of the battery. According to research findings, variations in electrode thickness affect the volume changes of the battery, thereby influencing the internal resistance and lithium-ion deposition patterns. Appropriate electrode thickness helps maintain tight contact between battery components, enhancing the cycling stability and rate performance of the CR2032 coin cell. However, excessively thick electrodes may lead to poor contact between the active material and the current collector, increasing internal resistance and compromising high-current discharge performance.
(2) Sealing pressure plays a vital role in the assembly process of the CR2032 coin cell. Proper sealing pressure ensures the integrity of the internal structure and the stability of electrical contact. Insufficient pressure may cause the electrode-electrolyte interface to lose contact, leading to rapid capacity decay and severely affecting the battery lifespan. Conversely, excessive pressure may lead to interface deterioration and increased resistance, or even damage the battery structure, posing potential safety risks.
(3) Baking conditions are essential for removing moisture from the electrodes. Optimal baking conditions help reduce the internal resistance of the CR2032 coin cell and improve its high-current discharge performance. Inadequate baking can result in residual moisture reacting with the electrolyte, leading to performance degradation. Heating in a vacuum environment can effectively remove internal moisture from the electrodes, enhancing both the performance and safety of the battery.
In summary, this paper attempts to enhance the electrochemical performance of lithium-ion batteries by precisely controlling and optimizing electrode thickness, sealing pressure, and baking conditions, thereby ensuring the stability and reliability of the CR2032 coin cell under high-current charge-discharge conditions.
3. Implementation of Countermeasures and Analysis of Results
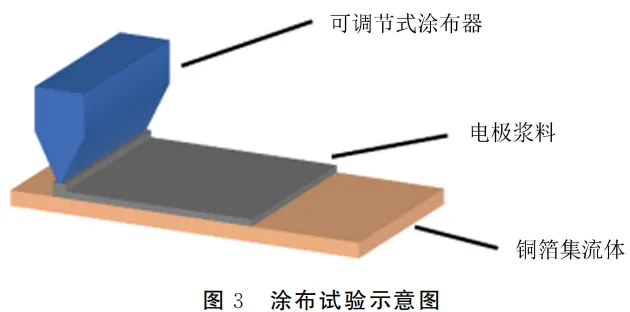
3.1 Electrode Coating Thickness Experiment
Electrode thickness significantly affects the kinetic performance of the battery. In this experiment, the thickness of the electrode slurry was adjusted by changing the gap of an adjustable coater. The schematic of this experiment is shown in Figure 3.
The thickness of the electrode plays a decisive role in the internal resistance and ion transport efficiency of the CR2032 coin cell. By precisely controlling the coating parameters, we evaluated how different thickness levels influence the high-current discharge stability of the CR2032 coin cell.
Four different coater thicknesses—50 μm, 100 μm, 150 μm, and 200 μm—were employed for electrode preparation. The corresponding average areal densities were 0.414mg/cm2、0.8845mg/cm2、1.7045mg/cm2、2.954mg/cm2, respectively. For each thickness, ten CR2032 coin cells were fabricated. The detailed experimental results are presented in Table 1.
Electrode Thickness Normal Cells / Total Cells Number of Cells Failing High-Current Discharge Percentage (%)
As the assembly of CR2032 coin cells is a manual process, operational errors may occur during assembly, leading to battery failure. Therefore, when calculating the percentage of cells unable to support high-current discharge, these failed CR2032 coin cells should be excluded.
The aforementioned experimental data reveals the influence of electrode thickness on the high-current discharge limitations (often referred to as the low-current discharge phenomenon) of CR2032 coin cells. Specifically, when the electrode thickness is 50 μm and 100 μm, the proportion of CR2032 coin cells failing to support high-current discharge is relatively low, at approximately 50%. This phenomenon is attributed to the tighter contact between the active material and the current collector in thinner electrodes, which reduces the internal resistance of the battery.
However, as the electrode thickness increases, particularly with the 200 μm electrodes, the failure rate for high-current discharge rises significantly to 100%. This trend indicates that excessively thick electrodes lead to poor contact between the active material and the current collector. Furthermore, during the charge-discharge cycles of the CR2032 coin cell, the active material becomes more prone to detaching from the current collector, further increasing the internal resistance. Therefore, optimizing electrode thickness is of great significance for reducing internal resistance and enhancing the performance of CR2032 coin cells.
Although comparing different coating thicknesses revealed that thinner coatings can effectively reduce the failure rate, the experiment still observed that high-current discharge failure occurred across all electrode thicknesses during the fabrication of CR2032 coin cells. Since this phenomenon persists even in thinner electrodes and becomes more pronounced in thicker ones, it is necessary to further explore the root causes of these discharge limitations in CR2032 coin cells.
3.2 Experiment on Appropriate Sealing Pressure
Appropriate sealing pressure ensures the integrity of the internal structure and the stability of electrical contact within the battery. In this experiment, the pressure of the CR2032 coin cell sealing machine (Figure 4) was adjusted to investigate whether optimizing the contact between electrode components could resolve the issue of high-current discharge failure in CR2032 coin cells.

CR2032 coin cells fabricated with electrode thicknesses of 50 μm and 100 μm exhibited the lowest proportion of high-current discharge failure. However, at a thickness of 50 μm, the foil was exposed (foil exposure), and the weighing error was significant. Therefore, all subsequent experiments utilized electrodes with a thickness of 100 μm. Under identical conditions, ten CR2032 coin cells were fabricated for each sealing pressure—0.65T, 0.7T, 0.75T, and 0.8T—using the 100 μm electrodes. The experimental results are presented in Table 2.
Sealing Pressure (T) Normal Cells / Total Cells Number of Cells Failing High-Current Discharge Percentage (%)
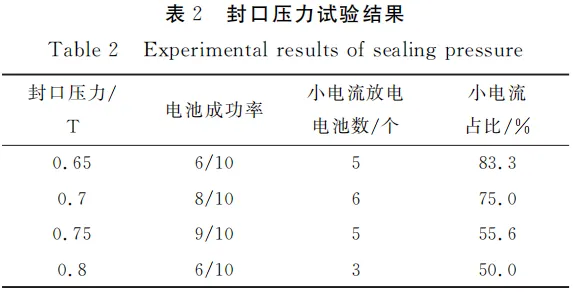
Experimental results indicate that excessively high or low sealing pressure severely affects the success rate of CR2032 coin cells. This is because insufficient pressure can lead to the loss of contact at the electrode-electrolyte interface, while excessive pressure may cause interface deterioration, increase internal resistance, or even damage the battery structure. As the sealing pressure increases, the proportion of CR2032 coin cells exhibiting low-current discharge initially decreases and then tends to stabilize.
Specifically, when the sealing pressure was increased from 0.65T to 0.75T, the percentage of CR2032 coin cells failing high-current discharge dropped significantly from 83.3% to 55.6%. This suggests that appropriate sealing pressure helps reduce internal micro-short circuits, thereby lowering the risk of discharge failure. However, when the pressure was further increased to 0.8T, although the failure rate slightly decreased to 50.0%, the overall success rate of the CR2032 coin cells declined. This may be due to internal structural deformation or damage caused by excessive pressure, which adversely affects the overall performance.
Further analysis revealed that at a sealing pressure of 0.75T, the probability of discharge failure was similar to that at 0.8T, while the fabrication success rate was better maintained. This indicates that 0.75T achieves an optimal balance between reducing discharge risks and maintaining production efficiency for CR2032 coin cells. However, through the comparison of various sealing pressures, it was found that the fundamental phenomenon of high-current discharge failure did not improve significantly. Therefore, it is concluded that the sealing pressure is not the root cause of the discharge limitations in CR2032 coin cells.
3.3 Influence of Electrode Baking Temperature and Duration
Optimal baking conditions contribute to reducing the internal resistance of the battery and enhancing the high-current discharge performance. Based on the optimization of parameters from the electrode coating thickness and sealing pressure experiments, a coating thickness of 100 μm and a sealing pressure of 0.75T were selected for subsequent tests. Using baking temperature as the variable, two temperatures—90°C and 120°C—were designed to investigate the causes of high-current discharge failure in CR2032 coin cells.
Two electrodes were first pre-baked in an oven at 90°C for 6 hours, and then subjected to vacuum baking at 90°C and 120°C, respectively, for 12 hours. Under identical conditions, CR2032 coin cells were fabricated using electrodes from these two baking scenarios, with 10 units per group. The experimental results are presented in Table 3. The electrodes from the previous experiment (baked at 80°C, with 0.75T sealing pressure and 100 μm coating thickness) were used as the reference group.
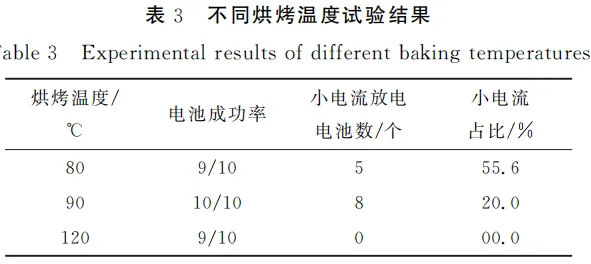
Based on the experimental data, we observed that the CR2032 coin cells prepared under elevated baking temperatures exhibited a higher success rate, and the phenomenon of high-current discharge failure was significantly suppressed. The discharge voltage profiles are shown in Figure 5. Specifically, under vacuum baking conditions at 90°C, the proportion of CR2032 coin cells failing high-current discharge dropped to 20%, whereas when the baking temperature was further increased to 120°C, this proportion decreased to zero.
These results indicate that as the baking temperature increases, moisture within the battery is removed more effectively, thereby reducing electrolyte side reactions caused by residual moisture. These side reactions typically increase the internal resistance of the electrode. Further analysis shows that when the baking temperature was raised to 90°C, the significant decrease in the failure rate for high-current discharge did not have a negative impact on the success rate of the CR2032 coin cells. At 120°C, internal moisture was more thoroughly removed, completely eliminating the phenomenon of high-current discharge failure. This finding confirms the importance of high-temperature vacuum baking in removing internal moisture from CR2032 coin cells. If not removed, this moisture reacts with the electrolyte, leading to electrolyte consumption and increased internal resistance, which ultimately impairs the high-current discharge capability of the battery.
4. Conclusion
Taking all experimental results into comprehensive consideration, we can conclude that inadequate baking during the fabrication process leads to residual moisture within the electrodes. The side reactions between this residual moisture and the electrolyte are the primary cause of increased internal resistance in the battery. Therefore, by optimizing the baking conditions—specifically by increasing the baking temperature and ensuring a vacuum environment—the internal resistance of the CR2032 coin cell can be effectively reduced. This enhances high-current discharge performance and mitigates the occurrence of discharge failure.
Reference:
MO Zuxue, XIE Xuerui, JIANG Haowen.
Solution to the Issue of High-Current Discharge Failure During the Charge-Discharge Process of CR2032 Coin Cells [J].
Superhard Material Engineering, 2025, 37(5): 34-38.

