How to evaluate and improve the electrolyte wettability of lithium-ion batteries? First of all, let us understand the factors that affect electrolyte wettability.
Factors Affecting Electrolyte Wettability of lithium-ion batteries
Active Material Particle Size
Smaller active material particles have a larger specific surface area. A larger specific surface area means a larger contact area between the active material and the electrolyte, allowing the electrolyte to interact more fully with the surface of the active material. Therefore, the wetting effect is generally better than that of larger particles.
Areal Density
During electrode fabrication, a higher areal density means an increased loading of active material per unit area, which is often accompanied by a higher electrode compaction density. This leads to reduced porosity and smaller pore sizes inside the electrode. As a result, the electrolyte struggles to fully penetrate and wet the deep interior of the electrode.
Effect of Compaction Density on Electrolyte Wetting Efficiency
In lithium-ion battery manufacturing, coated electrodes are calendered. The compaction density determines the porosity, effective pore size, and tortuosity of the electrode, all of which are closely related to the electrolyte wetting rate.
Effect of Cell Structure on Electrolyte Wetting Efficiency
The direction of electrolyte penetration varies with different cell structures.
In wound cells, the electrolyte penetrates the cell interior from both ends, while in stacked cells, the electrolyte infiltrates from three directions.
Under the same dimensions and energy density, stacked cells have higher wetting efficiency than wound cells due to the additional infiltration direction.
Effect of Separator and Electrolyte on Wetting Performance
Polyolefin materials generally have poor wettability with electrolytes, which can negatively impact battery performance.
Surface Tension and Viscosity of Electrolyte
The surface tension and viscosity of the electrolyte are two significant factors affecting its wetting performance.
The wettability between the electrolyte and the separator can be improved by adding wetting agents, which are typically ionic or nonionic surfactants.
Wetting Rate of Different Materials
Wetting rate: Anode > Separator > Cathode electrode
Porosity: Anode is typically 40%~60%, while separator and cathode porosity is 30%~40%
Vacuum Conditions
Vacuum conditions mainly affect the efficiency and rate of electrolyte wetting.
Multiple vacuum extractions during the wetting process help improve the electrolyte wetting effect.
While vacuum conditions facilitate electrolyte diffusion, they also lower the boiling point of the electrolyte, increasing its evaporation rate.
Wetting Time
The longer the wetting time of a fluid in a porous medium, the more complete the wetting.
Since wetting time is related to both efficiency and production cost, a reasonable wetting time must be established.
Wetting Temperature
Increasing the wetting temperature can improve electrolyte wettability by enhancing electrolyte fluidity, increasing interfacial affinity, and promoting gas release.
Low temperatures, on the other hand, can adversely affect the wetting rate and completeness.

How to evaluate the electrolyte wettability of lithium-ion batteries?
1 Experiments
1.1 Experimental Materials
1.2 Experimental Formula
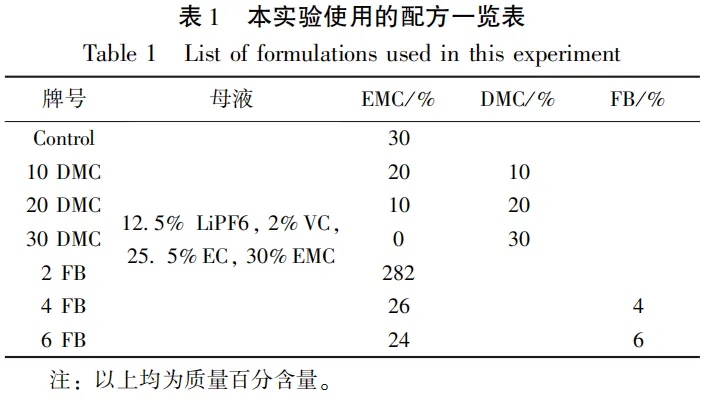
1.3 Experimental Instruments
- Electronic balance: Mettler Toledo
- Electrode hanging device: self-made
- Contact angle and surface tension meter: LSA100 Video Optical Contact Angle and Surface Tension Meter
1.4 Experimental Methods
Capillary Rise (Washburn) Method
The operating procedure for testing the electrolyte climbing height via the hanging method is as follows:
Place the device vertically and inject electrolyte into it, keeping the liquid level slightly lower than the height of the shell. Cut the samples to be tested into strips of 20 mm × 260 mm. The dimensions of the experimental platform are 55 mm (thickness) × 174 mm (length) × 200 mm (height); the recommended cutting length of the sample is ≥ 1.2 times the height of the shell.
Fix the sample in the grooves at the upper and lower ends of the partition using double-sided tape, ensuring the sample is straight and free of bending during fixation. Then slowly insert the partition with the fixed sample strip into the shell, and record the time and the corresponding scale of the liquid level. A scale line with an interval of 0.5 mm is designed on the left side of each groove on the partition, serving as the starting point of the wetting experiment. Then slowly remove the pipette and seal the injection hole with a rubber stopper. Subsequently, the height change ΔH of the electrolyte climbing in the electrode is recorded at regular intervals.
Wetting Balance Method
Contact Angle Method and Surface Tension Method
Cell Electrolyte Absorption Method
2 Results and Discussion
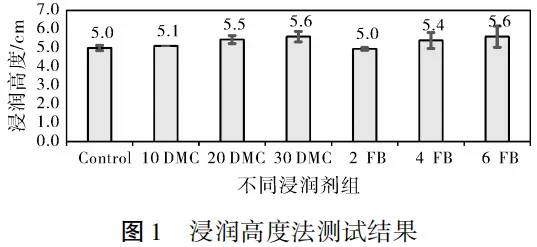
2.1 Wetting Height Method
Results Analysis:
The test results of the wetting height method are shown in Figure 1. As the addition amount of wetting agents FB and DMC increases, the wetting height increases, which is consistent with practical expectations. However, the numerical differences between different groups are small, the data deviation is large, the results are difficult to reproduce, and the consistency of the electrode sheets cannot be guaranteed. The possible reason is that there are many uncontrollable factors during the test, such as the uniformity of the electrolyte and the microstructure of the electrode surface, which may lead to large differences in the results of each test, making it difficult to obtain stable and reliable repeated results.
It is unable to accurately evaluate the actual wetting status:
Meanwhile, the wetting height method mainly evaluates the wetting performance based on the mass of electrolyte absorbed by the electrode within a certain period of time. However, this method may not fully reflect the true internal wetting condition of the electrode, especially for electrodes with uneven wetting or microstructural differences, which can lead to inaccurate evaluation results.
It is highly dependent on the size and shape of the electrode:
The test requires electrodes of fixed dimensions, which limits the applicability of this method to electrodes of different sizes and shapes. For non-standard-sized or specially shaped electrodes, additional processing and conversion may be required, increasing the complexity and uncertainty of the test.
This type of method has a long testing cycle:
To obtain relatively accurate evaluation results of wetting performance, the electrode usually needs to be immersed in the electrolyte for a period of time, which may result in a long test cycle. This is not conducive to rapid evaluation during R&D and production processes.
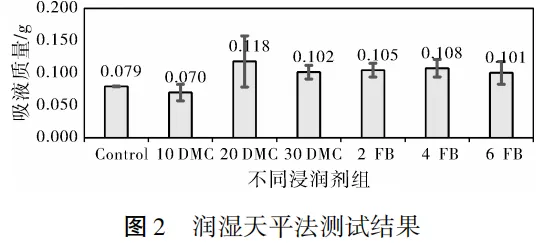
2.2 Wetting Balance Method
Results Analysis:
As can be seen from the wetting balance method test results, the overall numerical differences between different groups are small, with large data deviations and numerical anomalies. This makes it impossible to reflect the actual effect of wetting agents such as FB.
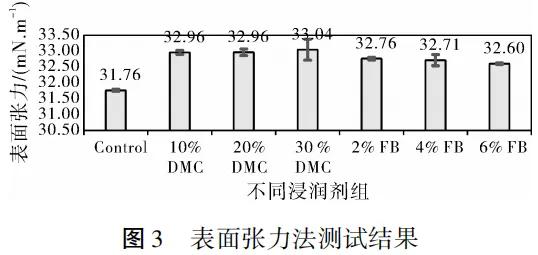
2.3 Surface tension method
Results Analysis:
The results of the surface tension method show that the surface tension of the electrolyte tends to increase after adding wetting agents. However, the overall numerical differences between different groups are small, with large data deviations, making it impossible to draw regular conclusions. The reason is that the wetting process of the electrolyte on the electrode involves not only interfacial interactions but also the siphon effect generated by the internal pores of the electrode.
2.4 Contact Angle Method
As shown in Figure 4, the contact angle method has two major problems. First, electrolyte droplets spread rapidly on the electrode surface and cannot remain stable. Second, it is difficult to fully unify the contact time and contact speed between liquid and electrodes during the sessile drop contact angle test, which makes the wetting time difficult to define. Meanwhile, as displayed in Figure 5, the contact angle cannot maintain relative stability for a certain period of time.
(1) Influence of surface roughness. Actual electrode surfaces are usually rough and inhomogeneous, causing contact angle hysteresis with inconsistent advancing and receding angles, which reduces test accuracy. Such surface inhomogeneity makes it hard to reflect the true wetting performance of electrodes.
(2) Uncertainty of electrode calendering quality. Poor calendering effect will lead to insufficient contact between droplets and the electrode surface during testing, lowering result reliability. For instance, powdery active materials may fall off or roll, resulting in unstable droplet morphology and inaccurate contact angle measurement.
(3) Operational complexity and measurement error. The contact angle test requires precise operation and professional skills. Multiple error factors including instrument precision, ambient temperature and humidity fluctuation will interfere with the final results.
(4) High dependence on electrode materials. Electrodes made of different materials present distinct surface properties. The contact angle method lacks sufficient adaptability for some special materials, so comprehensive evaluation combined with other testing methods is necessary.
2.5 Cell Liquid Absorption Method
It can be seen from the test results of the cell liquid absorption method that the liquid absorption capacity increases after adding wetting agents to the electrolyte. The cell liquid absorption rises with the increase of wetting agent dosage. In addition, the wetting effect of fluorobenzene is better than that of dimethyl carbonate (DMC). This method presents low overall test error and excellent repeatability, which is suitable for the evaluation of electrolyte wettability.
3 Conclusions
References
How to Improve electrolyte wettability of lithium-ion batteries?
- Electrolyte Injection Process
Improving the electrolyte injection process is a conventional method to enhance the wetting effect of lithium battery electrolytes. It can be optimized from multiple dimensions, including injection efficiency, injection conditions, standing time, and injection methods, so as to avoid insufficient wetting of the separator and electrode sheets by the electrolyte.
- Adding Electrolyte Wetting Agents
Adding additives to the lithium battery electrolyte can also improve its wettability. Wetting agents are essentially surfactants, which have the advantages of high surface activity, high thermal stability, low flammability, and high chemical stability. Adding wetting agents to the electrolyte can reduce the surface tension of the liquid, improve the wetting and penetration ability of the electrolyte on the electrode sheets, and thus enhance the electrochemical performance of the lithium battery.
- Reducing Electrolyte Viscosity
Increase the proportion of low-viscosity solvents (e.g., replacing EC/PC with DEC and EMC) to reduce the viscosity of the electrolyte. The decrease in viscosity is often accompanied by a decrease in surface tension (γ) (e.g., adding surfactants), which further reduces the solid-liquid contact angle (θ) and enhances the spreading ability.
- Improving the Microstructure of Electrode Sheets
Different compositions of active substances inside the electrode sheets will also affect the wetting effect of the electrolyte. The wetting efficiency can be improved by constructing a coating with a directional capillary structure. The main driving force for electrolyte wetting is capillary force; constructing microchannels perpendicular to the electrode sheets in the electrode coating can effectively improve the wetting efficiency and effect of the electrolyte.
