Lithium Dendrite Growth Mechanism and Suppression Methods
Lithium metal is considered the ideal anode material for next-generation high-energy-density batteries, such as Lithium-Sulfur and Lithium-Air batteries. This is due to its extremely high specific capacity, low reduction potential, and low density. However, the growth of lithium dendrites severely limits its practical application. Test your battery’s performance with Neware’s high-precision battery testing solution.
Lithium dendrites can pierce the separator, leading to short circuits. They also destroy the Solid Electrolyte Interphase (SEI) film. This results in the accumulation of “dead lithium,” capacity fade, and serious safety issues.
Today, we will discuss the growth mechanism, influencing factors, characterization techniques, and suppression strategies of lithium dendrites.

I. Growth Mechanism of Lithium Dendrite
1.1 What are the growth models of lithium dendrite?
Currently, the most widely accepted growth models for lithium dendrites include the following:
SEI Film Protection Model
Yamaki (1) and others proposed that the stress generated during lithium deposition causes the SEI film to rupture. Lithium then preferentially deposits at these defects to form lithium dendrites. Theoretically, the surface tension of the SEI film must reach a specific threshold to effectively suppress lithium dendrite growth.
(1) DOI: 10.1016/s0378-7753(98)00067-6.
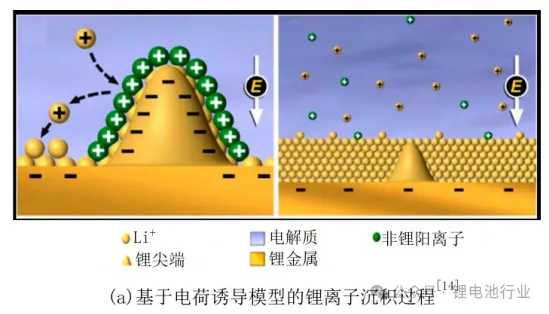
Charge Induction Model
Uneven charge distribution on the anode surface causes lithium ions to accumulate and deposit at protruding tips, forming lithium dendrites. By adding cations with low reduction potential, an electrostatic shielding layer can be formed at these tips to promote uniform lithium deposition.
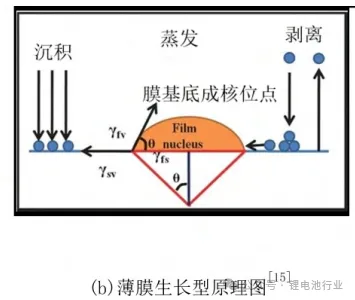
Thin Film Growth Model
Wang (2) and others pointed out that differences in surface energy and lattice structure between the deposited lithium film and the substrate induce lithium dendrite growth. Increasing the surface energy difference or using 3D current collectors can promote layered deposition.
(2) DOI: 10.1002/advs.201600168.
Other Models
In addition, there are models such as the heterogeneous nucleation model and the surface nucleation-diffusion model.
Summary
The growth of lithium dendrites is the result of multiple mechanisms acting together. Currently, no single model can fully explain all aspects of their behavior.
1.2 What are the factors affecting lithium dendrite growth?
- Temperature
SEI films formed at higher temperatures are more stable. This reduces interfacial impedance and promotes layered deposition, which improves cycling performance.
- Current Density
The morphology of dendrites at high current density differs from that at low current density. The magnitude of the current significantly affects the initial formation and growth patterns of lithium dendrites.
- Electrode Overpotential
High overpotential easily triggers lithium dendrite formation. Pulse charging can suppress their growth to some extent, while low overpotential is more favorable for cation diffusion.
- SEI Film
The stability and composition of the SEI film directly affect the uniformity of lithium deposition. Using additives or artificial SEI films can enhance its mechanical strength and stability.
- Deposition Substrate
Traditional current collectors, such as copper foil, often lead to local charge concentration. Substrates with a high specific surface area, uniform structure, and low nucleation energy barrier help promote uniform deposition.
1.3 Advanced characterization techniques for lithium dendrite
Conventional techniques struggle to monitor dendrite growth in real-time. In-situ characterization techniques provide dynamic and continuous observation of the lithium deposition process.
Methods such as Optical Microscopy, Electron Microscopy, Atomic Force Microscopy (AFM), and X-ray Photoelectron Spectroscopy (XPS) are essential.
These advanced tools help researchers deeply understand the mechanisms of dendrite nucleation and growth.

II. Suppression Methods for Lithium Dendrite
2.1 Stable deposition hosts and surface modification
3D Current Collectors
Porous copper mesh is a prime example. Its high specific surface area reduces local current density. This provides uniform sites for deposition and extends the cycling life of the battery.
Surface Modification
Using nitrogen-doped graphene as a conductive framework improves lithiophilicity. This promotes dendrite-free deposition. Even after two hundred cycles, the coulombic efficiency remains extremely high.
Materials such as tetrapyrrole-based polymers and graphdiyne are also effective. They use heteroatoms or intrinsic active sites to create strong interactions with lithium. This guides a uniform and stable deposition process.
2.2 SEI layer modification
Electrolyte Additives
Adding specific compounds to the electrolyte, such as lithium polysulfide and lithium nitrate, can form a stable SEI film. This ensures that no dendrites are generated even under high current densities.
Artificial SEI Film
Protective films, such as lithium fluoride layers prepared by magnetron sputtering, offer high ionic conductivity and mechanical strength. These films effectively promote uniform deposition. Using this method, the full cell cycling life can exceed five hundred cycles.
2.3 High-concentration and nano-structured electrolytes
High-Concentration Electrolytes
Using electrolytes with high salt concentrations reduces the amount of free solvent. This suppresses side reactions and enhances interfacial stability. However, the cost of these electrolytes is generally high.
Nano-structured and Anion-Tethered Electrolytes
Mixed electrolytes, such as ionic liquid-carbonate blends, offer high ionic conductivity and thermal stability. These materials effectively suppress dendrite growth and possess quasi-solid-state characteristics.
2.4 Solid-state electrolytes (SSEs)
Solid-State Electrolytes
Solid electrolytes possess high mechanical strength. This allows them to fundamentally prevent dendrites from piercing through.
Garnet-Type Electrolytes
These materials offer high ionic conductivity, but they often suffer from high interfacial resistance. Using thin alumina coatings prepared by atomic layer deposition can significantly improve the interface performance.
3D Nanofiber Networks
A three-dimensional network of nanofibers provides continuous ion channels. This structure enhances the stability of the polymer matrix and improves the overall battery performance.
2.5 Other approaches and emerging strategies
Separator Modification
Modifying the separator with materials such as graphene thin films can be highly effective. These films use regular channels to guide the uniform migration of lithium ions, which suppresses dendrite growth.
Multifunctional Separators
Self-standing cellulose nanofiber separators are a key example. These multifunctional separators can simultaneously inhibit the migration of polysulfides and the formation of lithium dendrites.
III. Conclusion
The commercial application of lithium metal batteries still faces severe challenges from dendrite growth. Current research has established a systematic framework covering growth mechanisms, influencing factors, characterization techniques, and suppression strategies. Methods such as 3D current collectors, SEI engineering, and high-concentration or solid-state electrolytes have shown positive results in practice.
Future Research Directions:
Multi-strategy Synergy Combining electrolyte modification, substrate design, and solid-state electrolytes can build a multi-layered protection system.
Cost and Process Optimization It is essential to reduce the manufacturing costs of high-salt electrolytes, nano-electrolytes, and solid-state materials to promote large-scale applications.
In-situ Characterization and Technical Integration Further development of in-situ techniques will reveal dendrite behavior under real battery operating conditions. This will guide better material and interface design.
New Material Development The potential of materials such as 2D porous structures and high polymers in controlling lithium deposition still needs deep exploration.
Summary
Only through interdisciplinary cooperation, mechanism research, and engineering optimization can we achieve high-safety, long-life lithium metal battery systems. This will drive their practical use in electric vehicles and large-scale energy storage.
Reference
DOI: 10.7540/j.ynu.20230403
Related News:
- Why does an unstable SEI film always form during battery testing? 2026 Update
- Lithium Ion vs Lithium Polymer: A Comprehensive Comparison Guide for 2024
- Sulfide-based All-Solid-State Batteries: Core Challenges and Breakthroughs in Materials and Interfaces 1
- Electrochemical Test Techniques for Lithium-ion Batteries: CV, EIS
