2 Successful Applications of Neware Battery Testers in Lithium-Sulfur Battery Research
Neware Battery Testers in Lithium-Sulfur Battery Research Case Study 1:
Developing a One‐Pot Strategy to Synthesize Metal–Covalent Organic Frameworks as Catalysts for Polysulfide Conversion and Ion Calibrators for Lithium Deposition

First Author: Ke Yang
Corresponding Author: Yibai He
Affiliation: Northwestern Polytechnical University (NWPU)
Equipment Used: Neware Battery Testing System (CT-4008T-5V10mA-164) for coin cells testing.
The latest version of Neware battery testing equipment for coin cells is CT-4008Q-5V50mA-HWX, contact us for specifications and a quote.
Research Background
In recent years, Lithium–sulfur batteries (LSBs) have been recognized as one of the most promising next-generation energy storage systems due to their high theoretical specific energy (2600 Wh·kg⁻¹) and the abundance of sulfur resources. However, their commercialization faces two major challenges: the “shuttle effect” of polysulfides, which leads to the loss of active materials and rapid capacity decay; and the growth of lithium dendrites, which poses safety risks and compromises cycling stability.
To address these issues, researchers have attempted to introduce functional materials into battery separators to catalyze polysulfide conversion and regulate lithium deposition behavior. Among these, Metal–Covalent Organic Frameworks (MCOFs) have emerged as a standout class of functional materials. MCOFs offer several key advantages:
i) They combine the tunable coordination metal sites of MOFs with the high crystallinity, chemical stability, and ordered porous structure of COFs;
ii) High Designability: Functional properties such as catalysis, adsorption, and charge distribution can be tuned by introducing specific metal nodes;
iii) Ideal for Separator Modification: Their excellent interfacial stability and ion selectivity significantly enhance the overall performance of LSBs.
Despite their potential, the application of MCOFs is currently limited by complex synthesis processes and difficulties in structural control—particularly the challenge of achieving single-atom metal doping.
Article Introduction
Recently, the team led by Associate Professor Yibai He from the School of Materials Science and Engineering at Northwestern Polytechnical University published a research paper titled “Developing a One‐Pot Strategy to Synthesize Metal–Covalent Organic Frameworks as Catalysts for Polysulfide Conversion and Ion Calibrators for Lithium Deposition” in the prestigious journal Advanced Functional Materials.
This work proposes a simple and efficient one-pot synthesis strategy to introduce Zn single atoms in situ into the COF framework. The resulting Zn-COF material possesses both high catalytic activity and superior ion-regulation capabilities. When applied as a coating for lithium-sulfur battery separators, it effectively suppresses the shuttle effect and lithium dendrite growth, leading to a significant leap in battery performance.
Core Highlights
Point 1: One-Pot Synthesis of Zn-COF
A one-pot reaction strategy was employed to achieve coordination between Zn2+ and the N/O atoms within the COF framework. The Zn exists stably as +2 valent single atoms, with a high metal content of 12.43%, exhibiting a stable structure and uniform distribution.
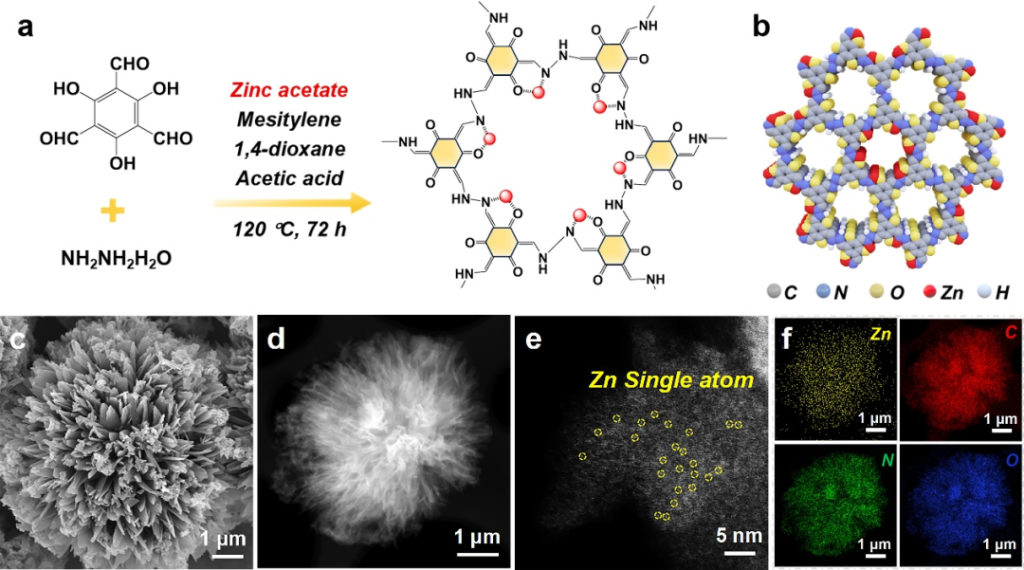
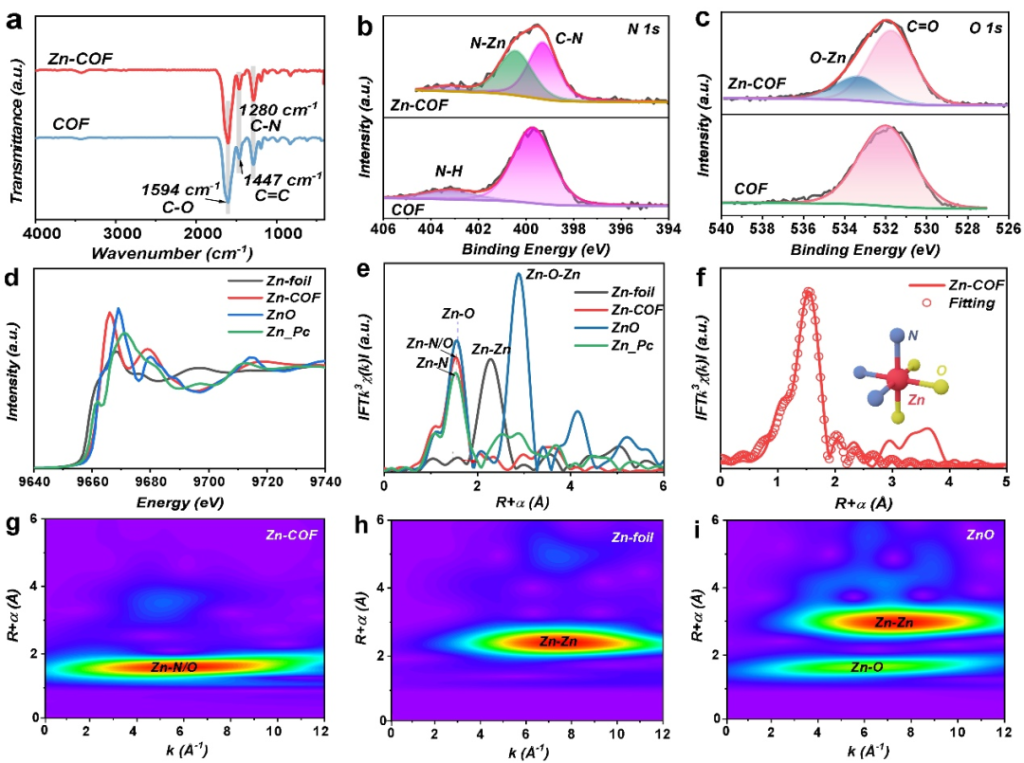
Point 2: Synergistic Catalysis and Adsorption: Enhancing Polysulfide Conversion
The Zn atomic sites effectively adsorb and catalyze the bidirectional conversion of Li2Sx species. Experimental data demonstrate that this significantly reduces polarization, enhances conversion kinetics, and suppresses the shuttle effect.
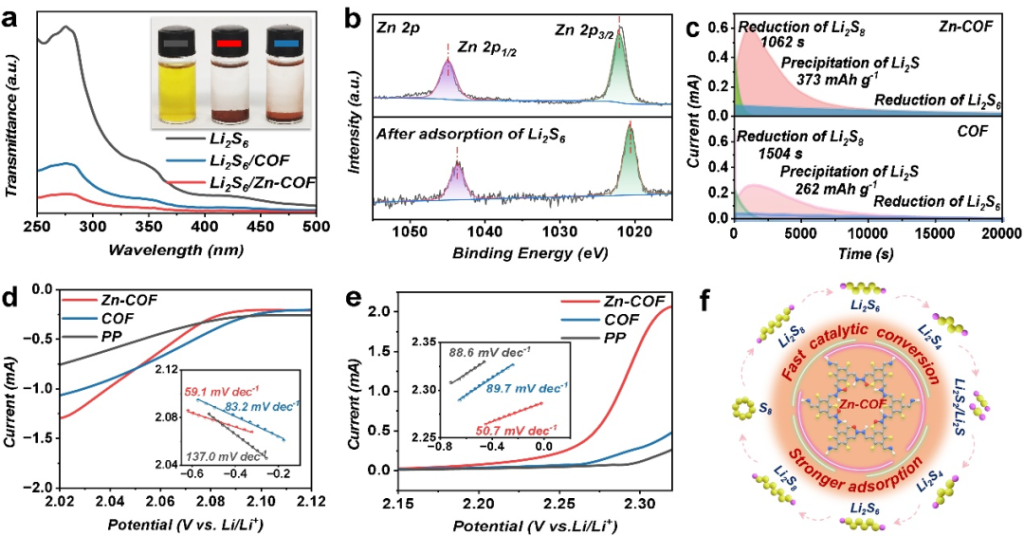
Point 3: Uniform Lithium Deposition and Effective Dendrite Suppression
The Zn sites exhibit lithiation-philic properties. Combined with the ordered porous structure, this achieves uniform Li-ion flux and balanced current density. The cycling life is extended to over 2,000 hours, with no dendrite formation even at a high current density of 10 mA·cm⁻².
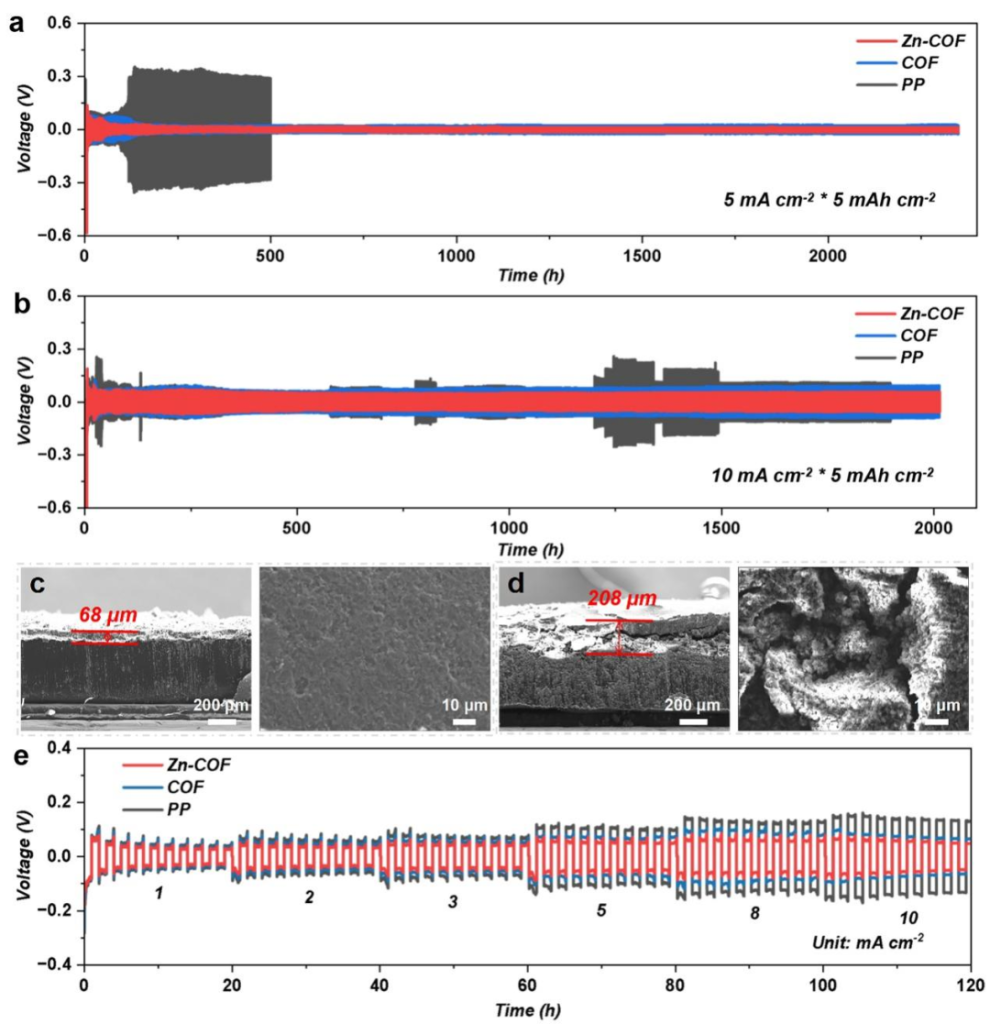

Point 4: Significant Leap in Li-S Battery Performance
The initial specific capacity reaches as high as 1535 mAh·g⁻¹ (at 0.1 C). At a high rate of 4 C, the battery maintains superior capacity retention over 1,000 cycles compared to benchmark materials. It remains stable under high sulfur loading and high areal capacity tests, demonstrating great potential for practical application.
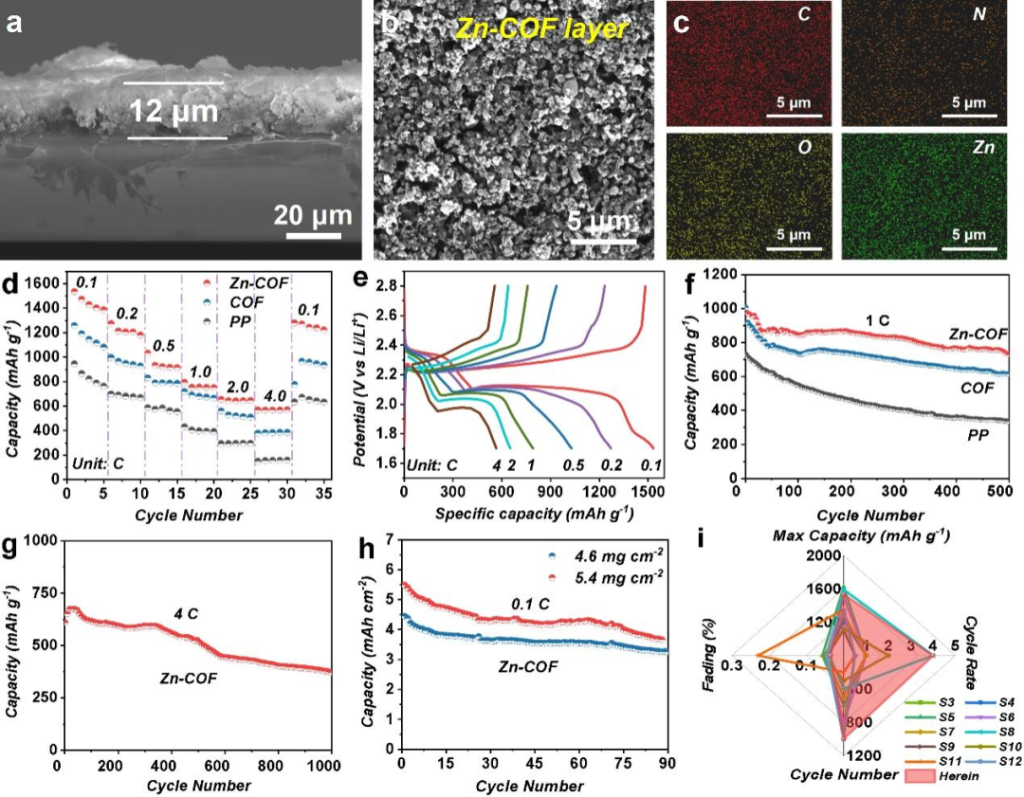
Conclusion
This research constructs a high-zinc-content Zn-COF via a one-pot method, achieving the dual functions of electrochemical catalysis and Li-ion regulation. It provides a feasible pathway for the practical application of Metal-COF (MCOF) materials in the field of Lithium-Sulfur batteries.
This strategy not only simplifies the synthesis process of single-atom metal COFs but also offers a universal approach for exploring other transition metal-doped MCOF materials, with the potential to be extended to various high-performance battery systems.
Ke Yang, Fei Zhao, Chan Li, Shaokun Zhou, Juan Li,Haoyuan Yang, Yichen Wang, Xuqing Liu, Yibo He*, Developing a One-Pot Strategy to Synthesize Metal–Covalent Organic Frameworks as Catalysts for Polysulfide Conversion and Ion Calibrators for Lithium Deposition. Adv. Func. Mater., 2025, 2501980.
Neware Battery Testers in Lithium-Sulfur Battery Research Case Study 2:
Expansion-tolerant architectures for stable cycling of ultrahigh-loading sulfur cathodes in lithium-sulfur batteries

Lithium-sulfur (Li-S) batteries hold immense potential, offering up to five times the energy density per unit weight compared to traditional lithium-ion batteries. However, they have long been hindered by significantly shorter lifespans. In a promising development recently reported in Science Advances, a research team at Monash University, led by Dr. Mahdokht Shaibani, has developed a stable new Li-S battery structure that could extend smartphone battery life to five days.
This new architecture enables unprecedented charge-discharge efficiency. Dr. Shaibani noted a persistent irony: the massive capacity of sulfur electrodes often leads to intense internal stress during operation, causing the components to crack and fail.
Specifically, stress-induced deformation and disconnection occur between the carbon matrix (which conducts electrons to the insulating sulfur) and the polymer binder (which holds the materials together), leading to rapid performance degradation. To address this, Shaibani and an international team moved away from dense bonding networks. Instead, they decided to “give the sulfur particles some breathing room.”
The new structure utilizes traditional binders but in a way that creates ultra-strong “bridge-like” bonds between the carbon matrix and sulfur particles. This configuration leaves extra space to accommodate the volume expansion that occurs during charging.
By using minimal amounts of binder between adjacent particles, the team created a network capable of absorbing structural changes and stress. In preliminary tests exceeding 200 cycles, the battery demonstrated a charge-discharge efficiency of over 99%. Researchers suggest this technology could enable smartphones to last five days or EVs to travel over 1,000 km on a single charge. The team has filed patents and plans to explore applications in EV power batteries and solar energy storage.
Related News:
- Prof. Yunhui Huang’s Group Leads the Way in Battery Innovation: Key Research Highlights (2025)-2
- Introduction to lithium-sulfur battery and lithium-sulfur electrolyte Published in 2026
- Lithium Ion vs Lithium Polymer: A Comprehensive Comparison Guide for 2024
- Prof. Yunhui Huang’s Group Leads the Way in Battery Innovation: Key Research Highlights (2025)-1
