The massive expansion of the EV market has pushed lithium-ion batteries beyond the 300 Wh/kg threshold, bringing costs down to nearly $100 per kWh. Despite this progress, conventional batteries with liquid electrolytes are hitting a plateau in terms of energy density and safety.
All-solid-state batteries are now seen as the core technology for the next generation of EVs, offering higher energy density, better safety, and longer life. In particular, sulfide-based all-solid-state batteries are gaining immense traction because their solid electrolytes can match the high ionic conductivity of liquid systems, making them a top research priority globally.
However, the practical application of sulfide-based ASSBs still faces multiple challenges. Technical bottlenecks spanning from materials to interfaces, and from composite electrodes to scale-up production, urgently need to be addressed. This first installment (Part I) will focus on the core material-level difficulties and the key sticking points of interfacial issues. By integrating the latest research progress, we will analyze the fundamental challenges and breakthrough directions for sulfide-based all-solid-state batteries.
Sulfide Solid Electrolytes for Sulfide-based All-Solid-State Batteries
1. Core Role and Performance
Sulfide solid electrolytes are the core components of sulfide-based all-solid-state batteries, providing lithium-ion transport channels and acting as a separator to prevent electronic short circuits between the anode and cathode. Since the discovery of Li10GeP2S12 in 2011, a series of high-ionic-conductivity sulfide solid electrolytes have emerged. These include the Li2S-P2S5 binary system, argyrodite-type Li6PS5X (X = Cl, Br, I), and thio-LISICON types. Their ionic conductivity can reach 1–25 mS/cm, approaching or even surpassing that of liquid electrolytes.
2. Challenge I: Poor Air Stability
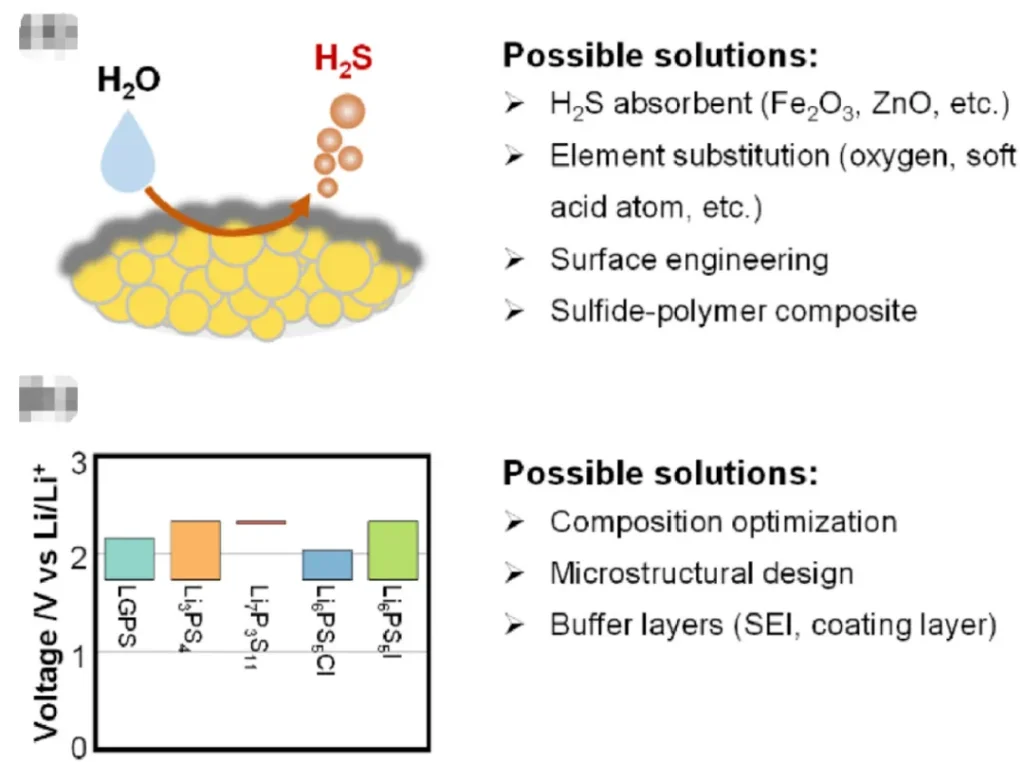
Specific Issues: Sulfide solid electrolytes react easily with moisture in the air to generate toxic hydrogen sulfide (H2S) gas. Simultaneously, the electrolyte itself converts into oxides, leading to a sharp decline in ionic conductivity. This phenomenon can be explained by the Hard and Soft Acids and Bases (HSAB) theory: phosphorus (a hard acid) in sulfides binds more readily with oxygen, while sulfur (a soft base) binds more easily with hydrogen to form H2S.
Optimization Strategies:
Additive Method: Adding metal oxides such as Fe2O3 or ZnO can absorb the generated H2S, but this cannot stop the hydrolysis of the electrolyte itself and may reduce ionic conductivity.
Element Substitution: Introducing oxygen or soft acid atoms like antimony (Sb), tin (Sn), or copper (Cu) can inhibit hydrolysis and enhance air stability. However, partial substitution may alter the crystal structure and sacrifice some ionic conductivity.
Surface Engineering: Constructing functional layers on the electrolyte surface to isolate moisture. For example, coating the surface of Li6PS5Cl with an oxysulfide nanolayer allows the electrolyte to maintain a high ionic conductivity of 2.50 mS/cm even after 30 minutes of air exposure.
Composite Systems: Building sulfide-polymer composite systems leverages the protective effect of polymers to improve air stability while enhancing the mechanical flexibility and processability of the electrolyte. Some composite systems can even withstand water immersion.
3. Challenge II: Narrow Electrochemical Stability Window
Specific Issues: The intrinsic electrochemical stability window of most sulfide solid electrolytes is 1.71–2.5 V (vs. Li/Li+). This fails to match commonly used high-voltage cathodes (above 4.0 V) and low-potential anodes (below 1.0 V). At high voltages, sulfides are easily oxidized into products like sulfites, phosphates, and sulfur. At low potentials, they may be reduced to Li2S, Li3P, etc., forming a high-impedance interfacial layer that hinders lithium-ion transport.
Optimization Strategies:
Structural and Compositional Optimization: Oxygen doping can enhance the electrochemical stability of Li6PS5X against lithium metal and oxide cathodes. Core-shell structure designs can broaden the electrochemical stability window of Li9.54Si1.74P1.44S11.7Cl0.3 to 0.7–3.1 V.
Buffer Layer Construction: Coating the cathode surface with oxides like LiNbO3 or LiTaO3, or constructing an artificial solid electrolyte interphase (SEI) on the anode surface, physically isolates the direct contact between the electrode and electrolyte to suppress interfacial reactions.
4. Progress in Scalable Synthesis
Mainstream synthesis methods currently include melt quenching, solid-state reaction, and wet chemical synthesis. Melt quenching requires high-temperature melting followed by rapid cooling, suitable for glass electrolytes but with high energy consumption. Solid-state reaction involves high-energy ball milling and annealing to produce crystalline electrolytes, though milling times are long and efficiency is low. Wet chemical synthesis uses solvents to mix precursors, shortening processing time and facilitating mass production, provided appropriate solvents are chosen to avoid sulfide degradation. Currently, companies like Mitsui Mining & Smelting (Japan) and POSCO (South Korea) have established pilot plants for sulfide solid electrolytes with annual capacities of 10 tons and 24 tons, respectively, verifying the feasibility of large-scale production.
Cathode and Anode Material Challenges and Optimization for Sulfide-based All-Solid-State Batteries
01 Cathode Materials for Sulfide-based All-Solid-State Batteries
Material Selection Requirements: To achieve high energy density, sulfide-based all-solid-state batteries must be paired with high-voltage and high-capacity cathode materials, such as nickel-rich or lithium-rich layered materials. While these materials have demonstrated high capacity advantages in liquid electrolyte systems, they face more prominent mechanical degradation issues in all-solid-state batteries.
Causes of Mechanical Degradation: In all-solid-state batteries, the contact between the cathode and the solid electrolyte is rigid, lacking the buffering effect provided by liquid electrolytes. Cathode materials undergo volume changes during the lithium insertion/extraction process. For instance, nickel-rich NCM can experience a volume contraction of 2%–8% during charging; the higher the nickel content, the more pronounced the volume change. This volume fluctuation leads to cathode particle cracking and the formation of voids between the particles and the electrolyte, which destroys lithium-ion and electron transport channels, ultimately causing capacity decay.
Optimization Strategies:
Microstructural Design: Optimizing the arrangement of primary grains within secondary particles—such as using radially oriented rod-like grains—can alleviate internal stress caused by anisotropic expansion and contraction, thereby reducing particle cracking. Single-crystal cathode materials, with their superior structural integrity and enhanced oxidative stability, effectively inhibit void formation and particle pulverization during cycling. They have been proven to significantly improve the cycling stability of sulfide-based ASSBs (though synthesizing completely boundary-free single-crystal cathodes remains a technical challenge, as most reported products still contain some boundaries and may still experience mechanical degradation after long-term cycling).
Particle Size and Shape Control: Small-sized cathode particles can shorten lithium-ion diffusion paths, reduce absolute volume changes, and mitigate contact loss with the electrolyte. However, the particle size ratio between the cathode and electrolyte must be balanced to maintain continuous ionic and electronic transport channels. Spherical particles help avoid local stress concentration caused by non-uniform intercalation, reducing mechanical degradation, whereas non-spherical particles may trigger uneven surface intercalation and accelerate performance decline.
02 Anode Materials for Sulfide-based All-Solid-State Batteries
Silicon-based Anodes:
Performance Advantages and Issues: The theoretical capacity reaches as high as 4200 mAh/g, more than ten times that of graphite. However, volume expansion during lithiation exceeds 300%, leading to particle pulverization and loss of contact with the electrolyte. Additionally, intense interfacial reactions result in low Coulombic efficiency.
Optimization Strategies: Size optimization (nano-silicon particles undergo amorphization during cycling to form a dense and uniform morphology that buffers volume expansion, while increasing the contact area with the electrolyte and conductive agents to enhance transport kinetics); practical exploration of micron-silicon (applying external pressure to suppress micron-silicon pulverization, bringing its cycling stability close to that of nano-silicon; constructing carbon-free high-loading micron-silicon anodes by limiting the contact area between the sulfide electrolyte and micron-silicon, achieving a loading of 5 mAh/cm² with 80% capacity retention after 500 cycles); technology migration (strategies used in liquid batteries, such as surface coating, core-shell structure design, and silicon-carbon composites, are expected to be migrated to all-solid-state systems).
Lithium Metal Anodes:
Performance Advantages and Issues: With a theoretical capacity of 3860 mAh/g and the lowest electrode potential, lithium metal is an ideal anode material. However, it faces two major challenges: interfacial instability and lithium dendrite growth. Chemical reactions occur when lithium metal contacts sulfide electrolytes, forming an interphase; some interphases have poor mechanical strength and crack as the lithium metal volume changes, leading to continuous lithium consumption and declining Coulombic efficiency. Furthermore, contact loss and uneven current density distribution induce lithium dendrite growth, which can pierce the electrolyte layer and trigger internal short circuits.
Anode-free Design:
Advantages and Issues: This design eliminates the lithium metal, reducing costs and safety risks, but it faces even more severe challenges, such as massive volume changes and intense interfacial reactions.
Optimization Case: An Ag-C (silver-carbon) nanocomposite layer can regulate lithium deposition behavior. Silver nanoparticles alloy with lithium to induce uniform deposition, while the carbon layer acts as a buffer to prevent direct contact between the lithium and the sulfide electrolyte. Pouch cells using this design have achieved energy densities exceeding 900 Wh/L with stable performance after 1000 cycles. However, the high cost of silver limits large-scale application, making the search for low-cost alternative materials a future priority.
Interfacial Issues and Mitigation Strategies in Sulfide-based All-Solid-State Batteries
1. Central Importance of the Interface
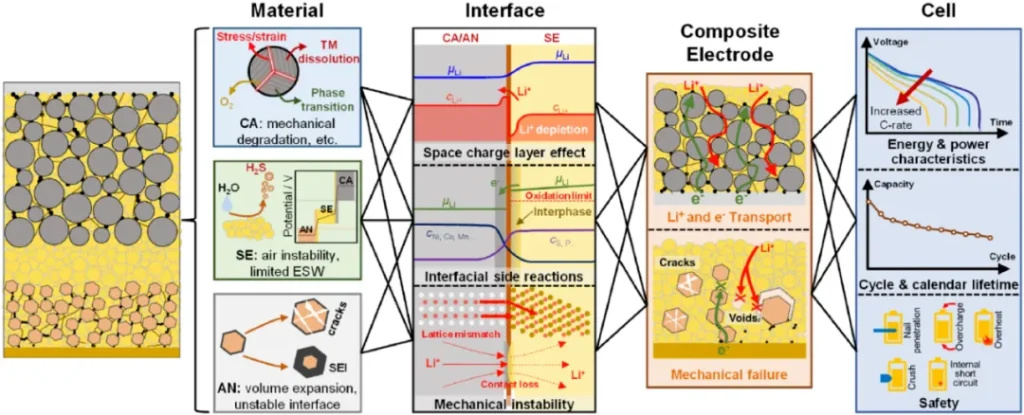
The interface in sulfide-based all-solid-state batteries is the critical conduit for ion and electron transport, as well as the primary source of performance degradation. Incompatibilities between electrodes and electrolytes in electrical, electrochemical, chemical, and mechanical aspects lead to the formation of space-charge layers, interfacial reactions, and mechanical instability. These three factors collectively restrict battery performance.
2. Issue I: Space-Charge Layer Effect
Formation Mechanism: Typically forms at the interface between oxide cathodes and sulfide electrolytes. Due to the difference in lithium chemical potential between the two, lithium ions migrate from the sulfide electrolyte to the oxide cathode, creating a lithium-ion depletion layer on the electrolyte side. To maintain charge neutrality, electrons migrate accordingly, triggering an interfacial potential shift.
Impact on Performance: The lithium-ion depletion layer significantly reduces local ionic conductivity and increases interfacial impedance, hindering transport. In experiments, the space-charge layer causes an extra oxidation plateau during early charging, similar to capacitor charging behavior. De-lithiation only occurs when the electrode potential reaches the cathode’s redox potential and the lithium-ion electrochemical potential is sufficient to overcome the energy barrier. This layer persists in subsequent cycles, continuously affecting transport efficiency.
Mitigation Strategies: Introducing coatings to suppress the space-charge layer—for example, inserting a LiNbO3 coating between a LiCoO2 cathode and a sulfide electrolyte—creates a smooth, matched interface that inhibits lithium-ion adsorption and reduces layer growth. DFT calculations show that LiNbO3 coatings lower the migration barrier for lithium ions while suppressing ion mixing and electrolyte oxidation. Another innovative design involves discontinuous coating of ferroelectric BaTiO3 nanoparticles on the cathode surface; their permanent reverse electric dipoles redistribute lithium ions and improve interfacial migration kinetics.
3. Issue II: Interfacial Reactions
Classification and Causes: Interfacial reactions include electrochemical and chemical types, both originating from the electrochemical instability and chemical reactivity of sulfide electrolytes. Electrochemically, the narrow stability window causes sulfides to be oxidized at the cathode and reduced at the anode. Chemically, elemental interdiffusion between the electrode and electrolyte leads to mutual decomposition, forming an interphase.
Impact on Performance: Most of these interphases have lower ionic conductivity than the sulfide electrolyte, increasing impedance. Some interphases are both ionically and electronically conductive, triggering continuous electrolyte decomposition and thickening of the interphase, which leads to rapid capacity decay.
Specific Manifestations:
Cathode-Electrolyte Interface: Sulfide electrolytes containing PS4 groups are easily oxidized by high-voltage oxide cathodes, generating PO4 groups and transition metal sulfides, accompanied by the interdiffusion of Co and P. One study observed a 10 nm thick interphase at the LiCoO2/Li2S-P2S5 interface with Co, P, and S diffusion. At high voltages, oxidation may also produce corrosive SO2 gas, threatening safety. However, some reactions are self-limiting, producing electronically insulating interphases that maintain certain cycling stability.
Anode-Electrolyte Interface: Reaction products depend on the electrolyte composition. Phosphorus-only sulfides (e.g., Li3PS4, Li7P3S11) react with lithium metal to form Li2S and Li3P; these are ionically conductive but electronically insulating, forming a passivation layer. Sulfides containing multi-valent metals like germanium, silicon, or tin (e.g., Li10GeP2S12) react with lithium to form alloys. These interphases are both ionically and electronically conductive, causing continuous reactions and interphase thickening, eventually leading to battery failure.
4. Issue III: Mechanical Instability
Formation Mechanism: Interfaces in all-solid-state batteries are rigid contacts. Volume changes in electrodes during cycling lead to lattice mismatch, contact loss, cracks, and void formation. This instability destroys transport channels and triggers performance decline.
Manifestations and Impacts:
Lattice Mismatch: Differences in lattice parameters create disordered layers at the interface, increasing diffusion resistance. Coherent interfaces allow fast transport but are extremely rare in sulfide systems. MD simulations show that non-coherent structures at the Li-metal/LLZO interface form disordered lithium layers and voids, leading to failure.
Contact Loss: A more widespread issue; electrode volume changes destroy tight contact with the electrolyte, forming voids. This concentrates current at limited contact points, increasing local current density and stress, which further cracks the materials in a vicious cycle. During de-lithiation at the lithium anode, if lithium removal is faster than replenishment, voids form and contact area decreases. During subsequent deposition, this induces dendrite growth and internal short circuits. While stack pressure can maintain contact via creep, excessive pressure forces lithium into grain boundaries or pores, promoting dendrites. One study found that increasing stack pressure shortened cycle life, with mechanical shorts occurring under high pressure even without applied current.
Real-world Case: At the interface of nickel-rich materials and beta-Li3PS4 electrolytes, significant contact loss and a large increase in void volume appear after 50 cycles, leading to rising impedance and capacity decay.
Mitigation Strategies: Single-crystal cathodes and sulfide electrolytes with good mechanical flexibility alleviate contact loss through their structural advantages. Proper fabrication and stack pressure can also effectively improve interfacial contact and battery stability.
Neware all in one for Sulfide-based All-Solid-State Batteries
Integrate the battery cyclers and battery test molds into a temperature chamber (0–60°C).

Contact us to learn more. NEWARE
More:
YouTube video
All Solid-State Batteries: from Sulfide-based Electrolyte to Halide-based Electrolyte
