Supercapacitors, Capacitors VS Batteries
Source: WeChat Official Account “Learn Batteries Together”
I. Charging and Discharging Principle of Supercapacitors:
The difference between a capacitor and a battery:
1. Working Principle
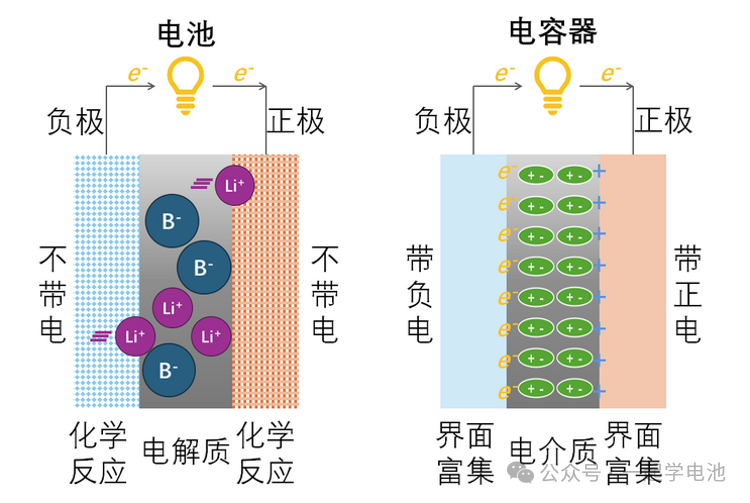
Capacitor: Stores energy through an electric field, with charge accumulating between the electrodes and the dielectric.
Battery: Stores energy through chemical reactions, which occur during charging and discharging.
2. Energy Density
Capacitor: Low energy density, suitable for short-duration energy release.
Battery: High energy density, suitable for long-duration energy supply.
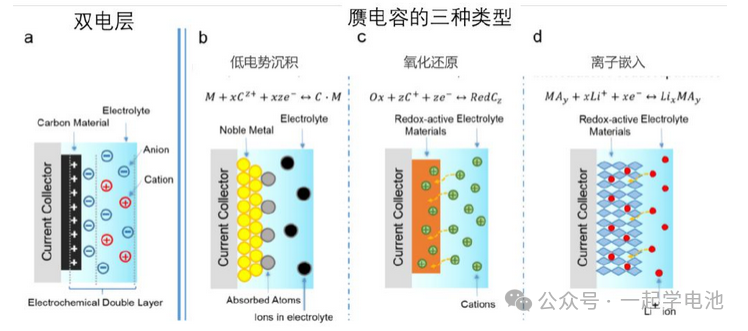
The defining characteristic of supercapacitors is the use of an electrolyte containing anions and cations—similar to that used in batteries—to replace the dielectric material found in conventional capacitors.
When a supercapacitor is fully charged, the negative electrode carries a negative charge while the positive electrode carries a positive charge. However, unlike a conventional capacitor where the internal dielectric molecules can only orient and polarize to create a counter-electric field, the supercapacitor utilizes an electrolyte with mobile ions. Consequently, cations in the electrolyte accumulate on the negative electrode side, forming an “Electric Double Layer” (EDL) with the negative plate. Similarly, anions migrate to the positive electrode side, forming another “Electric Double Layer” with the positive plate. Each of these double layers can be viewed as an individual “capacitor” formed between the migrated ions and the electrode.
Three common principles of “Pseudocapacitance”:
Underpotential Deposition (UPD): Ions from the electrolyte are adsorbed onto the electrode surface at a relatively low potential, undergoing spontaneous reduction to provide additional charge accumulation.
Redox Reactions at the Electrode Interface: Electrode materials such as ruthenium dioxide (RuO2) and manganese dioxide (MnO2) can change their valence states by gaining or losing electrons, allowing the material surface to carry additional charge.
Ion Intercalation: An intercalation method similar to the principle of lithium-ion batteries, where ions are inserted into the electrode structure to store extra charge.
All three methods involve redox reactions on the electrode surface, which are categorized as chemical reactions.
Supercapacitors cannot replace batteries or conventional capacitors; instead, they serve as an intermediate solution that combines the characteristics of both.
In laboratory environments, the energy density of supercapacitors can reach 40Wh/kg, which already exceeds that of lead-acid batteries, though a significant gap remains compared to lithium-ion batteries. In terms of performance, they integrate the features of both batteries and capacitors, offering unique advantages such as extremely high instantaneous power density, exceptionally long cycle life, and a wide operating temperature range. However, they also face challenges, including low peak energy density (relative to batteries), low overall energy density, and low operating voltage.
II. Composition of Supercapacitors
1. Electrode Materials
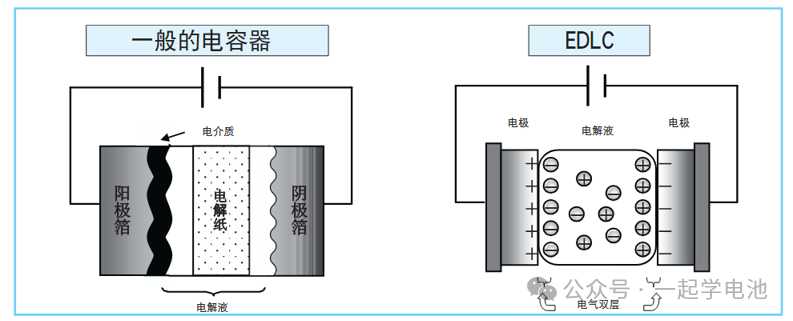
Common electrode materials for supercapacitors include porous carbon, conducting polymers, metal oxides, metal sulfides, and MXenes. Currently, commercial supercapacitors are dominated by porous carbon-based materials, which account for over 85% of the global market. Carbon materials possess a high specific surface area (1000–2500 m²/g) and excellent electrical conductivity. These properties enable the formation of a stable electric double-layer structure at the electrode-electrolyte interface, facilitating rapid ion adsorption and desorption, making them the standard configuration for modern electric double-layer capacitors (EDLCs).
Requirements for Electrode Materials:
High specific surface area and abundant reaction sites to ensure high capacity.
Low electron transport resistance to ensure high electrical conductivity.
Rapid electrolyte penetration into micropores and easy ion access to the material’s interior to enhance rate performance.
High ionic and electronic conductivity.
Chemical and structural stability for long cycle life.
Abundant material availability and simple preparation processes to reduce costs.
2. Electrolytes
Common electrolyte types for supercapacitors include aqueous electrolytes, organic electrolytes, ionic liquid electrolytes, and solid-state/gel electrolytes. Among these, aqueous electrolytes are safe and widely used; however, they decompose easily at high operating potentials.
| Characteristic | Aqueous System | Organic System | Ionic Liquid System | Gel/Solid-State |
| Voltage Range | Low | High | Highest | Moderate |
| Safety | High | Medium | High | High |
| Cost | Low | Medium | High | Low – Medium |
| Applications | Experiments, Low-voltage devices | Mainstream Commercialization | High-end Energy Storage | Wearable / Flexible Electronics |
Requirements for Electrolytes:
Wide electrochemical stability window to prevent decomposition under high voltage.
Good chemical stability and non-corrosive to electrodes.
Low viscosity and low cost with high industrial feasibility.
High ionic conductivity for rapid charge-discharge rates and high power density.
3. Comparison Between Supercapacitors and Lithium-ion Batteries
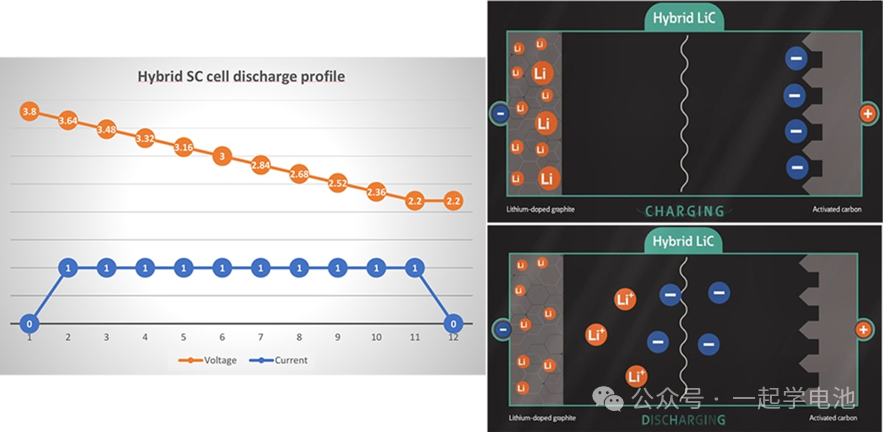
Supercapacitors exhibit a voltage drop during the discharge process (unstable power output), as they primarily store energy through physical electrostatic adsorption rather than chemical reactions. Compared to batteries, they possess a significantly higher number of charge/discharge cycles and a higher discharge rate.
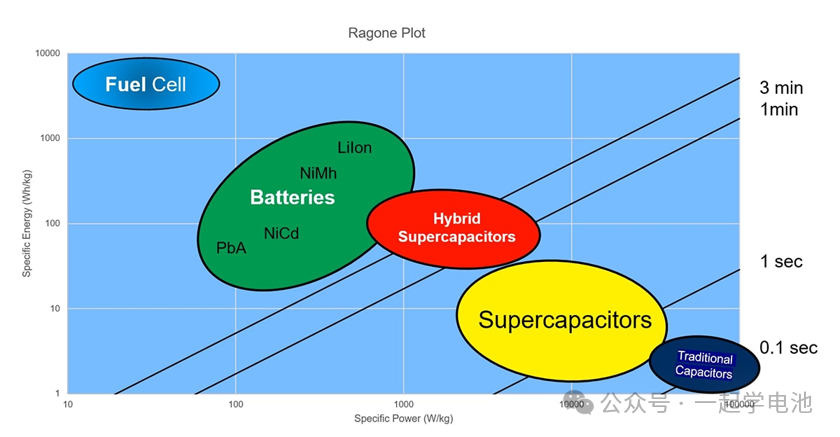
Devices with high energy density but low power density are located in the upper-left corner; these include fuel cells and batteries. Conversely, devices with high power density but low energy density, such as conventional capacitors and supercapacitors, occupy the lower-right corner. Hybrid supercapacitors fall between these two groups. The black diagonal lines in the figure represent operating time: supercapacitors operate for several seconds, hybrid capacitors for several minutes, and batteries for several hours or longer.
| Performance Parameters (性能参数) | Aluminum Electrolytic Capacitor (铝电解电容) | EDLC (双电层超容) | Hybrid Supercapacitor (混合超容) | Lithium-ion Battery (锂离子电池) |
| Energy Density (能量密度) | 0.01 ~ 0.3 Wh/kg | 1.5 ~ 8 Wh/kg | 4 ~ 80 Wh/kg | 100 ~ 265 Wh/kg |
| Power Density (功率密度) | > 100 kW/kg | 5 ~ 50 kW/kg | 3 ~ 10 kW/kg | 1 ~ 3 kW/kg |
| Temperature Range (温度范围) | -40 ~ +125 ℃ | -40 ~ +85 ℃ | -40 ~ +70 ℃ | -20 ~ +60 ℃ |
| Cycle Life (可充放电次数) | Unlimited (无限) | 100k ~ 1M times (10~100万次) | 20k ~ 100k times (2~10万次) | 500 ~ 15k times (0.05~1.5万次) |
| Service Life at RT (室温下工作寿命) | > 20 years | 5 ~ 15 years | 5 ~ 10 years | 3 ~ 5 years |
| Item (项目) | Supercapacitor (超级电容器) | Lithium-ion Battery (锂离子电池) | Sodium-ion Battery (钠离子电池) |
| Low-Temp Performance (低温性能) | Normal operation down to -40℃ (正常工作至-40℃) | Significant capacity decay below 0℃ (<0℃ 容量衰减明显) | Some systems down to -30℃ (部分体系可至-30℃) |
| High-Temp Performance (高温性能) | Stable up to 70–85℃ (稳定至 70-85℃) | Lifespan drops above 60℃ (>60℃ 寿命下降) | Slightly better than Li-ion (高温稳定性略优于锂电) |
| Safety (安全性) | No thermal runaway (无热失控) | Risk of thermal runaway (有热失控风险) | Slightly better than Li-ion but still needs protection (稍优于锂电但仍需防护) |
| Overall Performance (整体表现) | Widest temperature window (最宽温度窗口) | Requires strict thermal management (需严格温控) | Potential system (潜力型体系) |
In the past two years, the proportion of Lithium Iron Phosphate (LFP) batteries in newly manufactured electric vehicles in China has surpassed that of Nickel Manganese Cobalt (NMC) batteries. However, their startup performance under low-temperature conditions in northern winters remains a significant issue. Integrating a small supercapacitor to provide short-term auxiliary power is a potential solution.
The performance of supercapacitors depends on many complex factors: material composition, microstructure, electrolyte type, electrode thickness, pore size distribution, temperature, and scan rate, among others. Traditional experiments often require extensive trial and error, which is time-consuming and costly. Machine learning (ML) can “learn patterns” from existing data to predict performance, optimize designs, and guide experimental procedures. By employing machine learning methods, researchers can reduce experimental costs and screen for electrode materials that are both more affordable and higher-performing, potentially leading to a broader replacement of lithium-ion batteries.
III. Testing Objectives
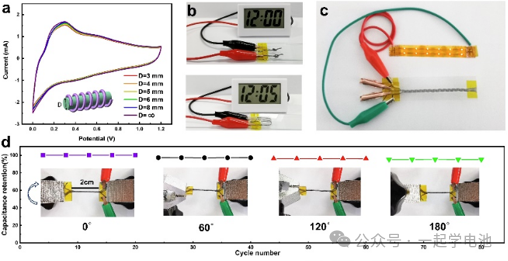
In the laboratory phase, supercapacitor testing covers a variety of cell and component configurations. A common practice involves coating the electrode slurry onto porous nickel foam, which is then dried and compressed to serve as the working electrode for testing. For batch samples, coin cell (button cell) formats are typically employed for long-term cycling tests. In contrast, flexible materials are assembled into thin-film or laminated structures to evaluate both their mechanical bending durability and electrochemical performance.
Commercial supercapacitors are primarily applied in energy storage modules, electric vehicle start-stop systems, rail transit energy recovery, and distributed energy regulation systems. Their packaging forms are mostly similar to those of lithium-ion batteries, including cylindrical, prismatic (square), and pouch (laminated) types. They can function as independent energy storage units or be combined with lithium-ion batteries to form hybrid energy storage systems (HESS).
Testing Objectives:
Energy Density: The amount of energy stored per unit mass or volume.
Power Density: The rate at which energy can be delivered.
Cycling Stability: The ability to maintain performance over hundreds of thousands of cycles.
Supercapacitor Performance Testing:
1. Cyclic Voltammetry (CV)
2. Galvanostatic Charge-Discharge (GCD)
3. Electrochemical Impedance Spectroscopy (EIS)
4. Cycle Life Testing
IV. Summary
Major Challenges in Supercapacitor Commercialization
Technical Challenges
Low Energy Density: Currently only 1/5 to 1/10 of lithium-ion batteries. Both volumetric and gravimetric energy densities struggle to meet long-range endurance requirements.
High Material Costs: High-performance carbon materials and metal oxides involve high preparation costs and complex processes, making large-scale production difficult.
Limited Operating Voltage: The low decomposition voltage of aqueous electrolytes (1.2V) restricts further improvements in energy density.
Energy Retention and Leakage: Significant self-discharge issues lead to rapid energy decay during long-term storage.
Industry and Market Challenges
Vague Market Positioning: Caught between traditional capacitors and batteries, resulting in unclear application boundaries.
Incomplete Ecosystem: Standardization of systems and testing methodologies has not yet been unified.
High Application Costs: The cost per Watt-hour (Wh) remains higher than that of lithium-ion batteries.
System Discrepancies: Differences in charge-discharge characteristics compared to lithium batteries mean they cannot directly replace existing battery-powered hardware.
Potentials and Innovation Breakthroughs
Technical Innovation Directions
High-Energy Density Material Systems: Developing composite electrode materials (Carbon + Oxides, Carbon + Polymers) combined with novel ionic liquid electrolytes and aqueous hybrid capacitors.
Structural Design Optimization: Leveraging nanoporous structures, 3D electrodes, and flexible thin films.
Device Integration and Intelligence: Focusing on miniaturization, on-chip integration, and combining intelligent power modules with energy management chips.
Future Application Fields of Supercapacitors
New Energy Vehicles (NEVs): Synergizing with lithium-ion batteries to handle instantaneous acceleration and regenerative braking energy recovery.
Grid Energy Storage: Providing rapid response to voltage fluctuations and frequency regulation.
Consumer Electronics: Enabling ultra-fast charging or serving as emergency backup power supplies.
Wearable Devices: Utilizing flexible supercapacitors as a lightweight, durable alternative to traditional batteries.
Test your batteries and supercapacitors——> Neware battery testers