Top 5 Most Common Reference Electrodes in Electrochemistry
Source: WeChat Official Account “Electrochemistry and Electrocatalysis”
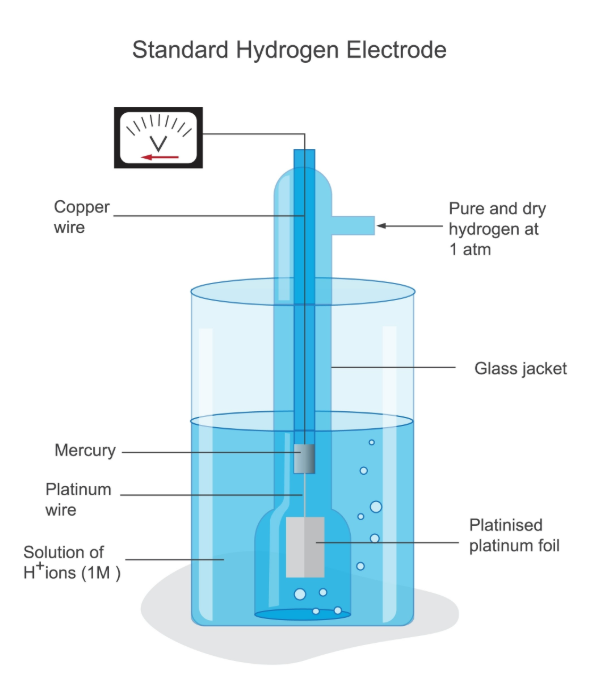
One of the 5 most common reference electrodes in electrochemistry: Standard hydrogen electrode (SHE / NHE)
Composition: A platinum sheet coated with platinum black is immersed in an acidic solution with a hydrogen ion activity of 1 mol/L, and pure hydrogen gas at a pressure of 1 atm is continuously introduced.
Characteristics: It is the absolute standard for all electrode potentials. Its standard electrode potential is arbitrarily defined as 0.000 V at any temperature.
Applicable Systems: Primarily used for theoretical research and calibration of other reference electrodes. Due to the need for a continuous supply of flammable hydrogen gas and the susceptibility of platinum electrodes to poisoning, it is rarely used directly in daily practical operations.
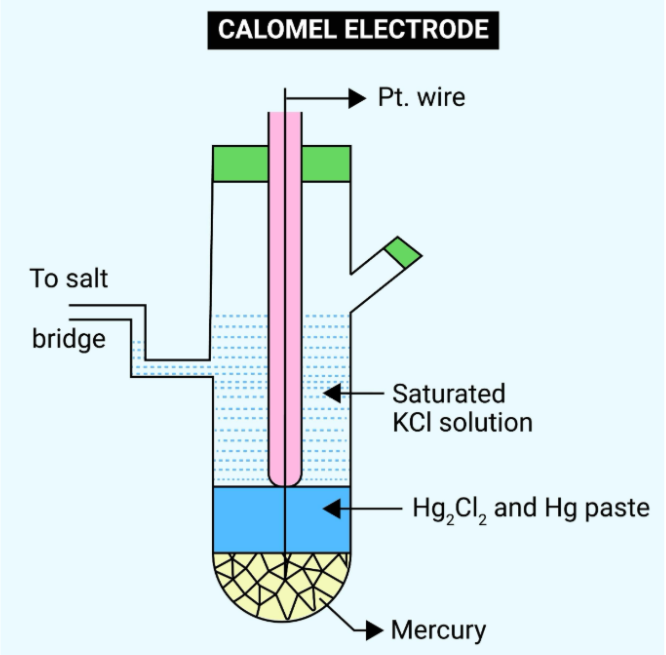
Two of the 5 most common reference electrodes in electrochemistry: Saturated calomel electrode (SCE)
Composition: Consists of metallic mercury (Hg), calomel (mercurous chloride, Hg₂Cl₂), and a saturated potassium chloride (KCl) solution.
Characteristics: Highly stable potential with good reproducibility. At 25°C, the potential relative to the standard hydrogen electrode is approximately +0.241 V.
Applicable Systems: Widely used in neutral and acidic aqueous solutions.
Precautions: Due to the presence of highly toxic mercury, and the potential instability caused by the disproportionation reaction of calomel above 80°C, it is gradually being replaced by Ag/AgCl electrodes in many fields. Furthermore, it contains chloride ions and is not suitable for systems sensitive to chloride ions.
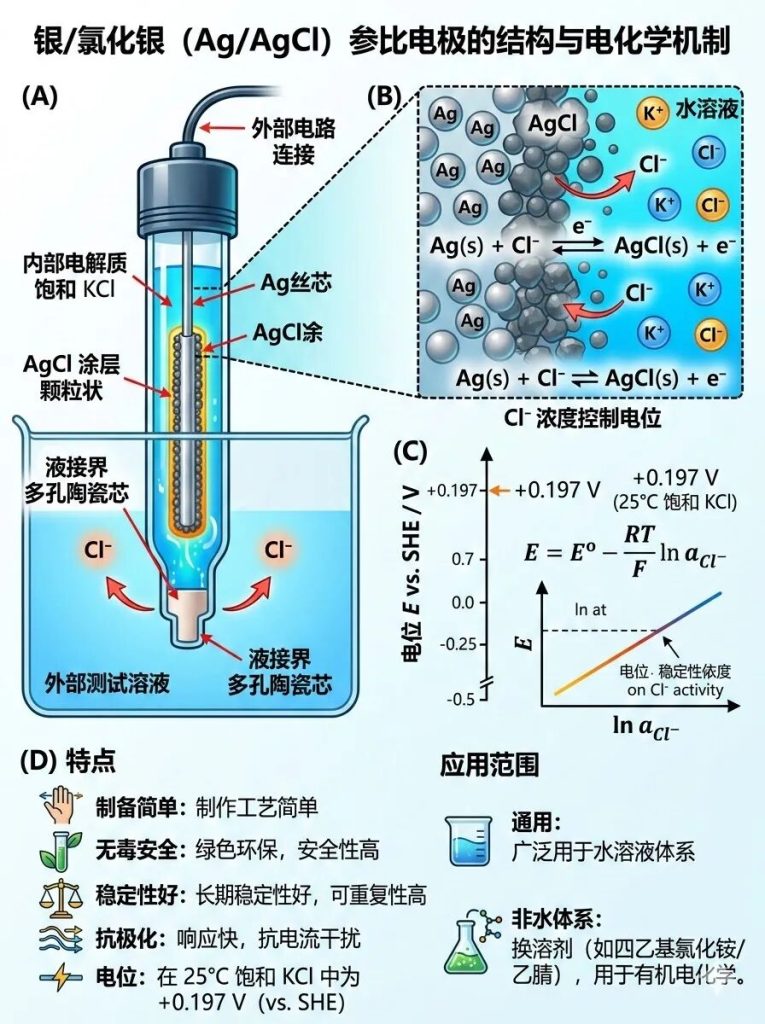
Three of the 5 most common reference electrodes in electrochemistry: Silver/silver chloride electrode (Ag/AgCl)
Composition: A silver wire coated with a layer of silver chloride (AgCl) is immersed in a potassium chloride (KCl) solution of known concentration (usually saturated, 3 M, or 1 M).
Characteristics: Simple to prepare, non-toxic, highly stable, and with strong resistance to polarization. At 25°C (in saturated KCl), its potential is approximately +0.197 V (vs. SHE).
Applicable Systems: Currently the most widely used reference electrode, extensively used in aqueous systems. By changing the internal solvent (e.g., using an acetonitrile solution of tetraethylammonium chloride), a reference electrode for non-aqueous systems can also be prepared.
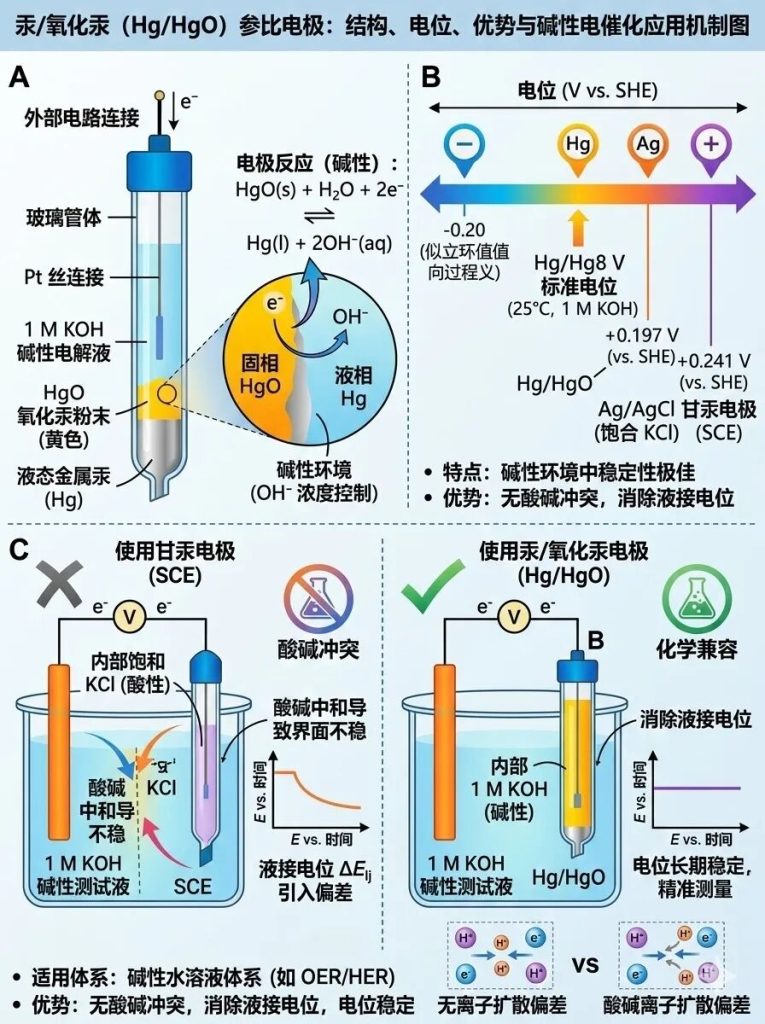
Four of the 5 most common reference electrodes in electrochemistry: Mercury/mercuric oxide electrode (Hg/HgO)
Composition: Consists of metallic mercury (Hg), mercuric oxide (HgO), and an alkaline solution (typically 1 M KOH or 1 M NaOH).
Characteristics: Extremely stable in alkaline environments. At 25°C (in 1 M KOH), its potential is approximately +0.098 V (vs. SHE).
Applicable Systems: Specifically designed for alkaline aqueous solutions (e.g., OER/HER testing in electrocatalysis).
Advantages: Its use in alkaline test solutions effectively eliminates the liquid junction potential, avoiding potential drift caused by acid-base neutralization as seen with saturated calomel electrodes.
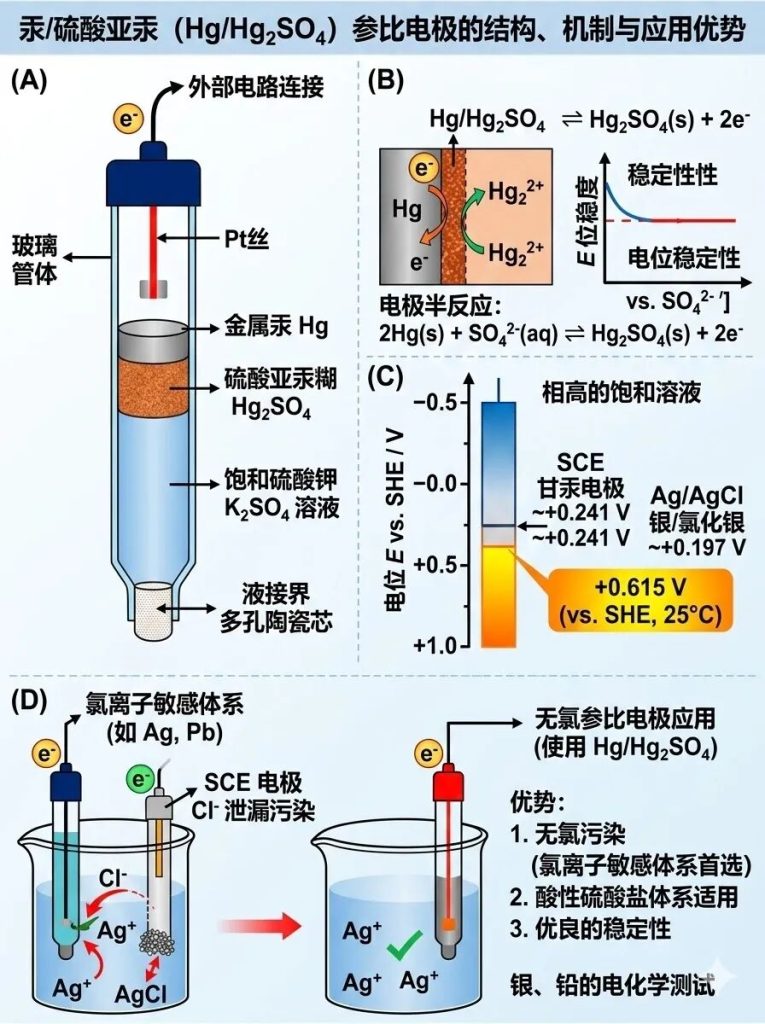
Five of the 5 most common reference electrodes in electrochemistry: Mercury/mercurous sulfate electrode (Hg/Hg₂SO₄)
Composition: Consists of metallic mercury (Hg), mercurous sulfate (Hg₂SO₄), and a saturated potassium sulfate (K₂SO₄) solution.
Characteristics: At 25°C, its potential is approximately +0.615 V (vs. SHE).
Applicable Systems: Primarily used in acidic sulfate systems.
Advantages: When your test system is highly sensitive to chloride ions (Cl⁻), or when the working electrode reacts with chloride ions (e.g., when testing the electrochemical behavior of silver and lead), this chloride-free reference electrode is typically chosen to prevent chloride ion leakage from the SCE or Ag/AgCl electrode and contamination of the solution.
While we strive for technical accuracy, errors or omissions may occur due to the evolving nature of battery technology. If you identify any inaccuracies or potential copyright concerns, please contact us via private message. We are committed to addressing such issues through immediate corrections or removal.
