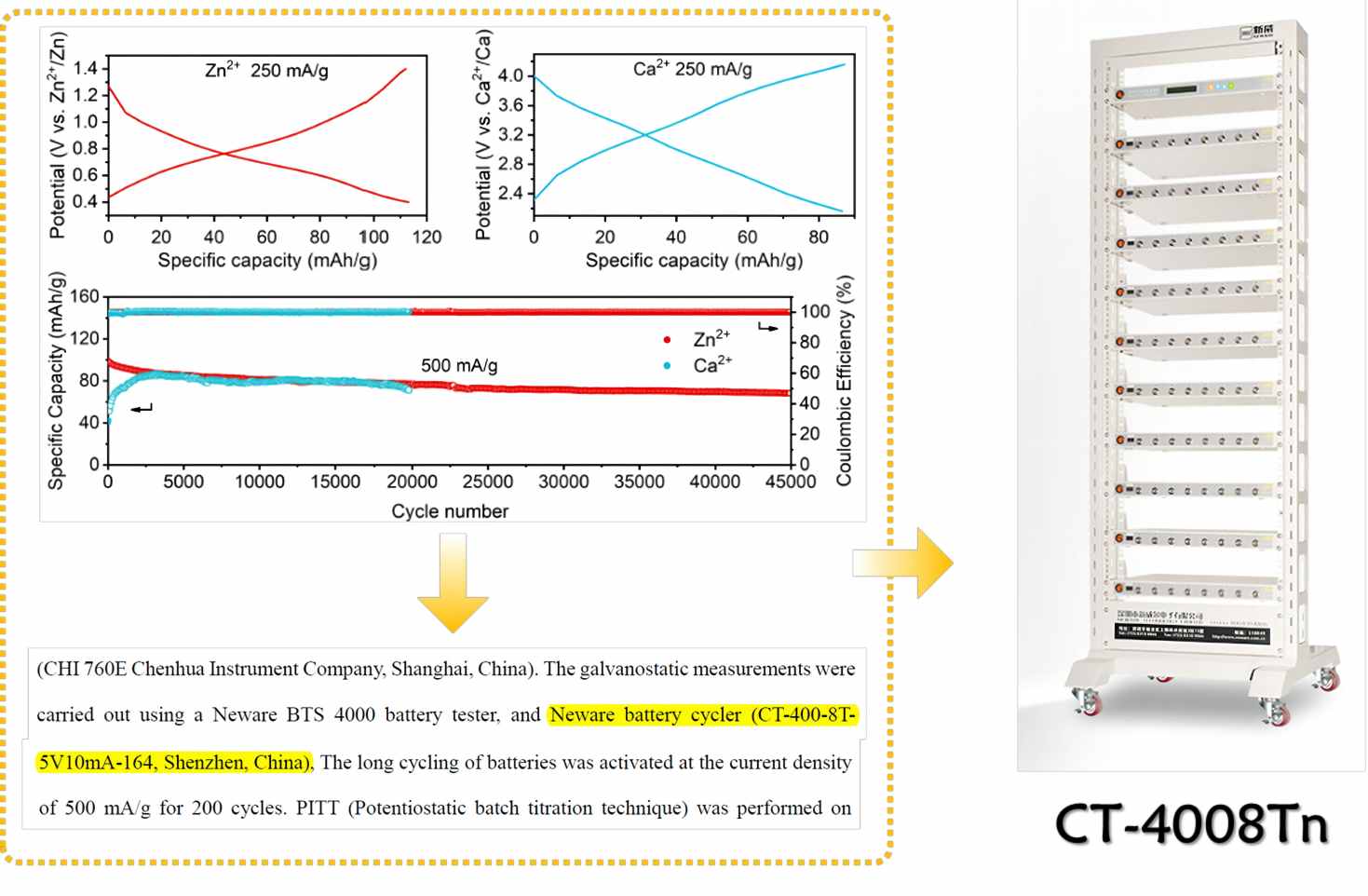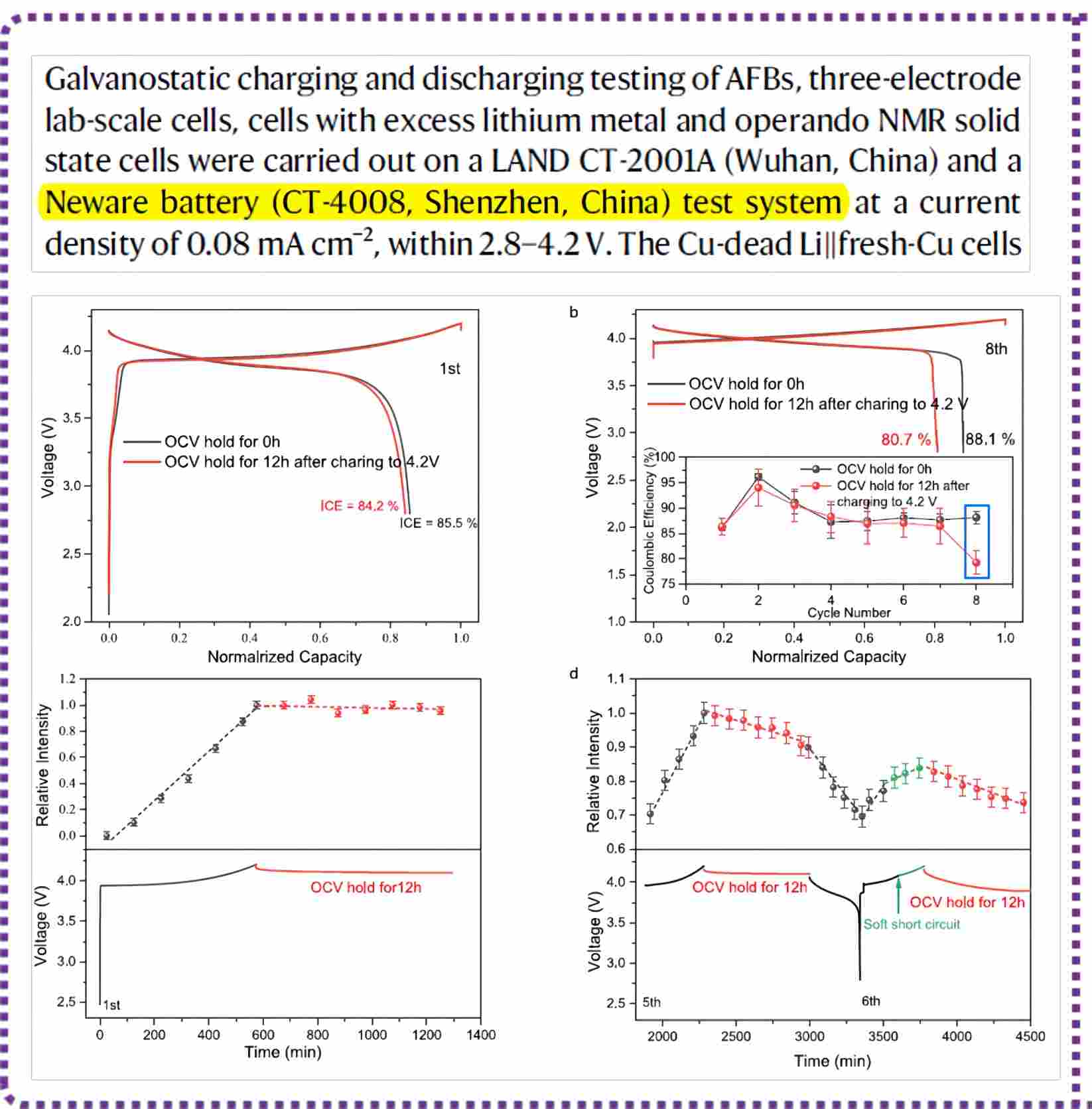
NEWARE Battery Testing System Empowers Solid State Battery Research 2024 post
Understanding the Failure Process of Sulfide-Based All-Solid-State Lithium Batteries via Operando Nuclear Magnetic Resonance Spectroscopy Primary author: Ziteng Liang (Xiamen University) Co-Primary author: Yuxuan Xiang (Xiamen University) Corresponding author: Yong Yang (Xiamen University) NEWARE Battery Testing System Empowers Solid State Battery Research Research achievement and introduction to the academic article Sulfide-based all-solid-state lithium batteries have attracted significant attention from the industry and academia due to their potential for high energy density and safety. However, in the case of lithium-negative electrode-type all-solid-state lithium batteries, non-active lithium is continuously generated during charge-discharge cycles, resulting in poor cycling performance and limiting their practical applications. Currently, non-active lithium consists of two parts: (1)