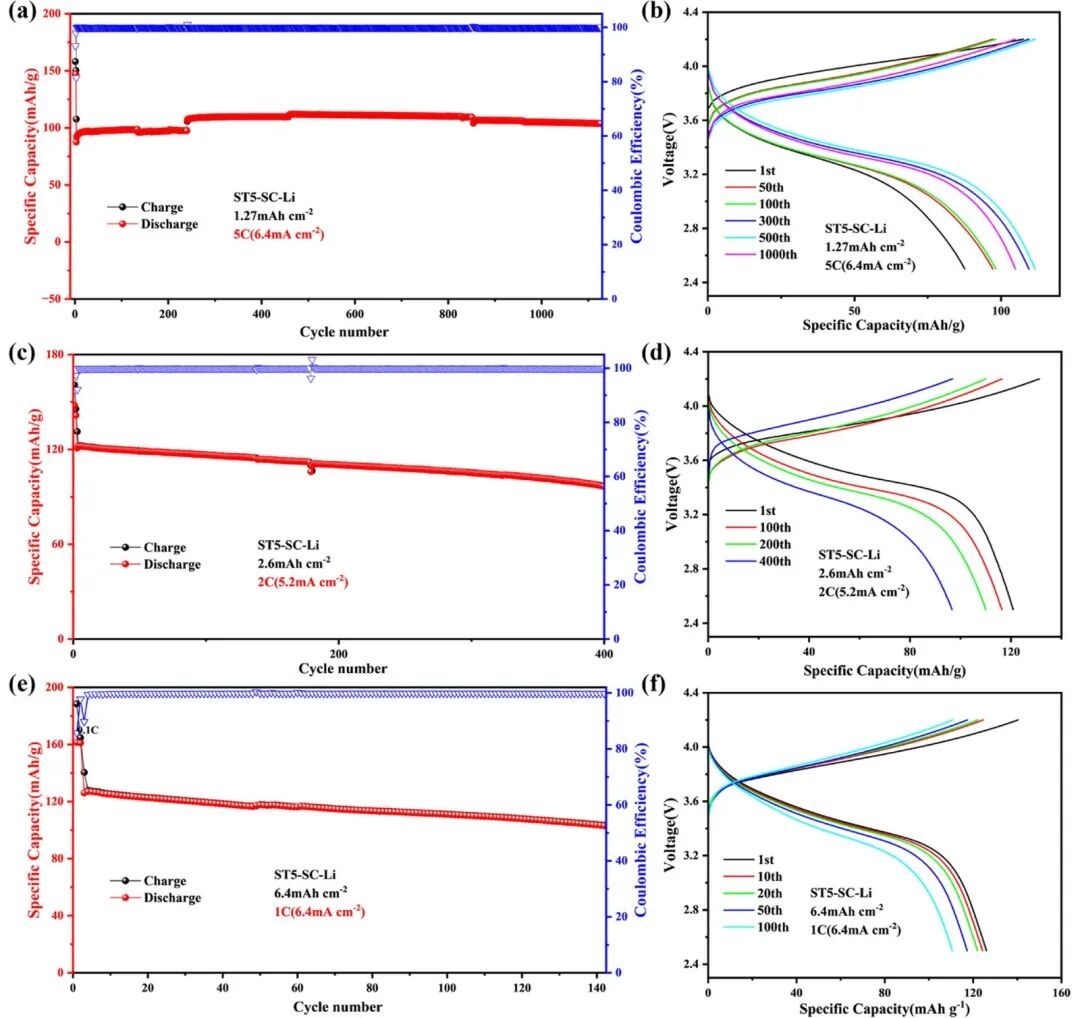
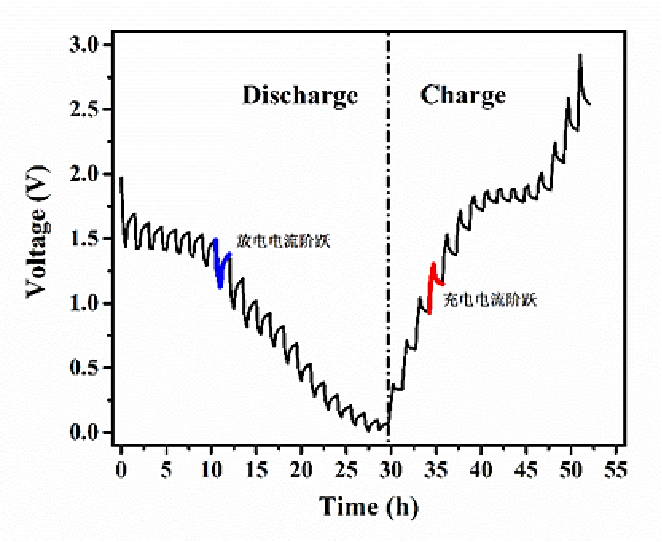
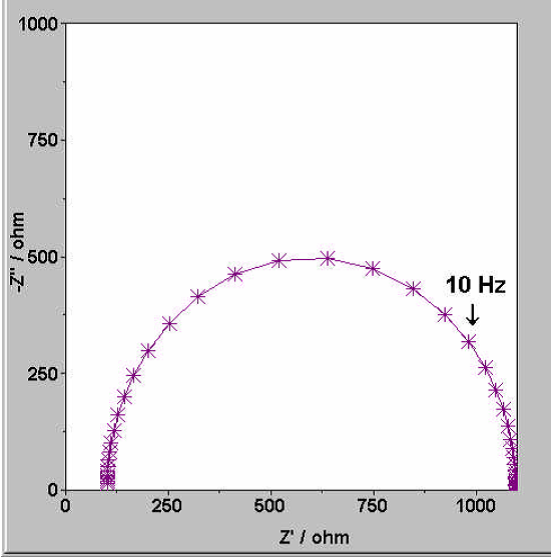
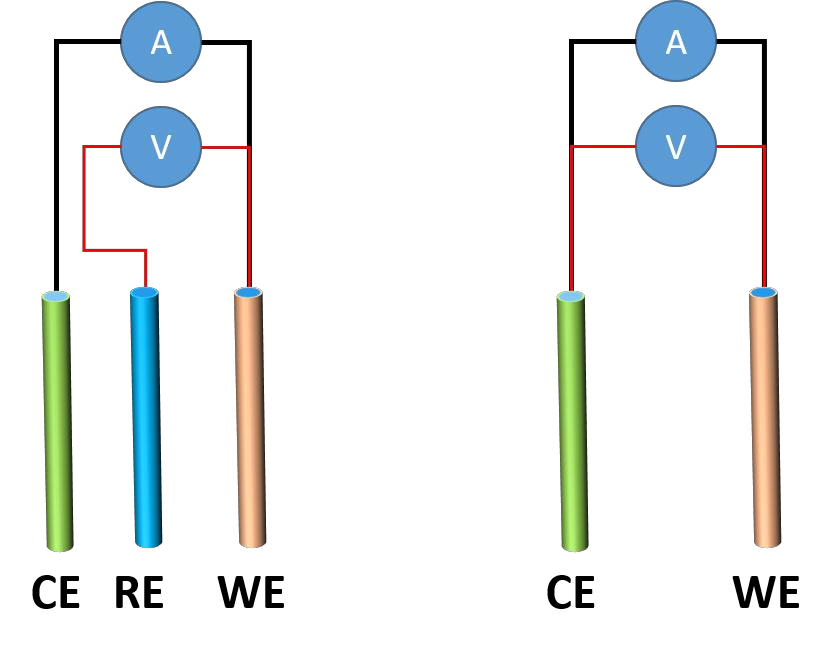
Battery test: Cyclic Voltammetry (CV), Working Condition Simulation, and Pulse Testing 2024
Battery test: Cyclic Voltammetry (CV), Working Condition Simulation, and Pulse Testing Cyclic voltammetry(CV) Cyclic voltammetry (CV) is an electrochemical measurement method that applies a triangular waveform pulse voltage to the closed loop formed by the working electrode and the counter electrode. The potential on the working electrode/electrolyte interface is systematically varied at a defined scan rate, inducing oxidation/reduction reactions of the active species present on the working electrode. This allows the measurement of the resulting current response during the electrochemical reaction. During the CV testing process, several issues may arise, and appropriate measures should be taken to address them: 1.Double-Layer Capacitance: At high scan rates, the capacitive