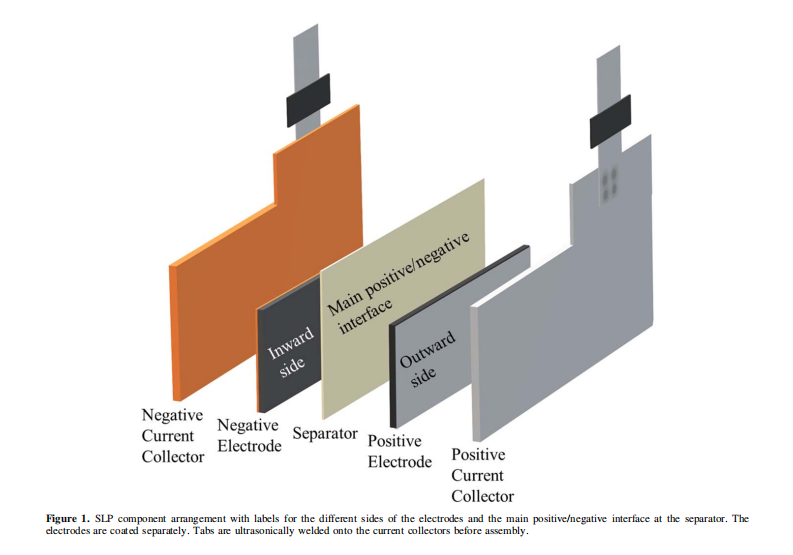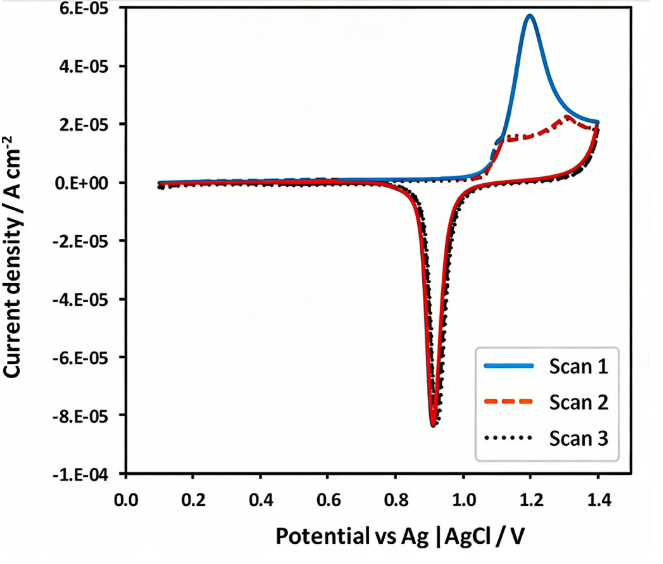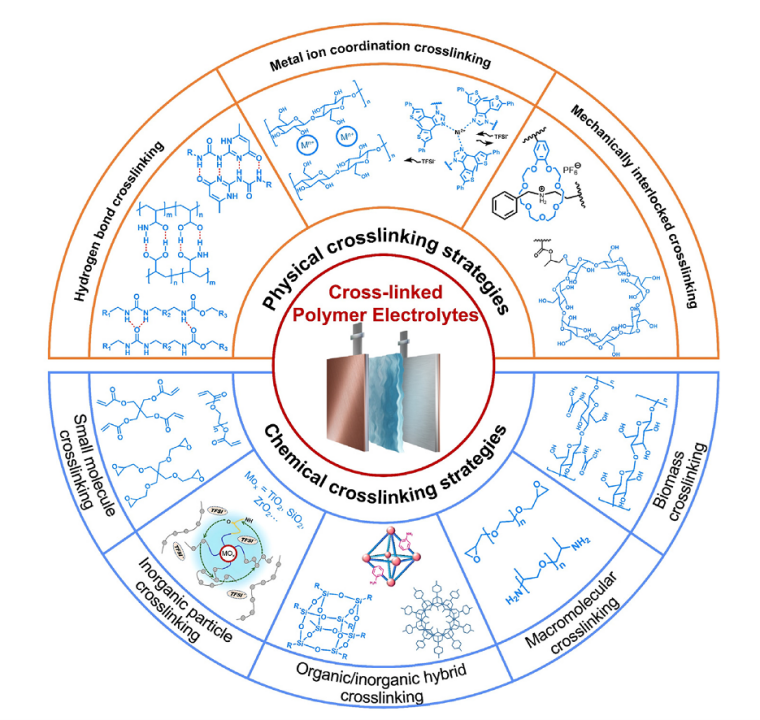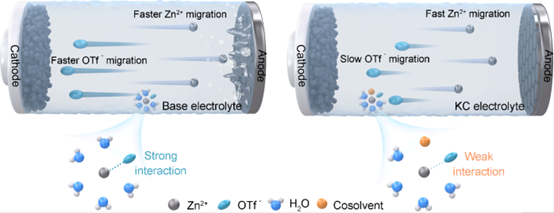
Design, Assembly, and Testing of Full Coin Cells: Tutorials and Case Studies 2026 post
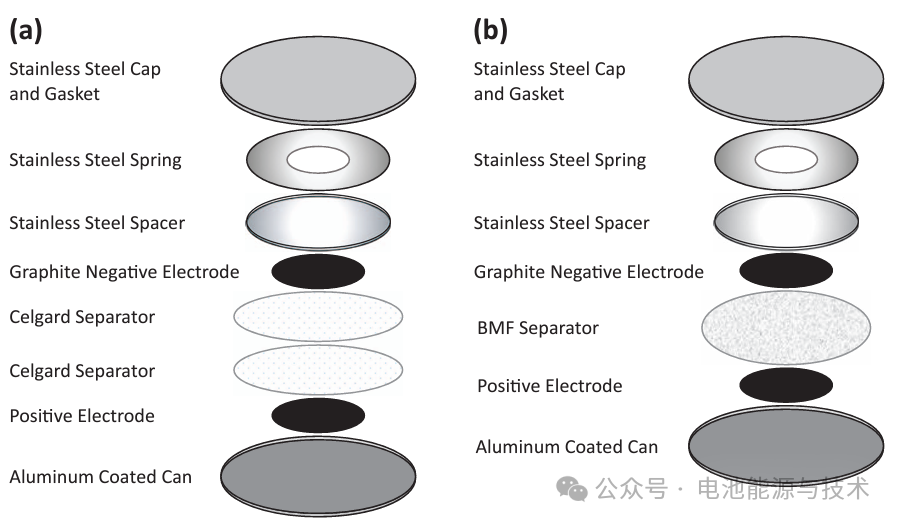
Design, Assembly, and Testing of Full Coin Cells: Tutorials and Case Studies 1. Introduction of Full Coin Cells A full cell is a complete battery system comprising a cathode, anode, separator, electrolyte, and casing. Unlike half-cells, full cells provide an accurate assessment of the electrochemical and mechanical performance of a battery under actual operating conditions. While a half-cell typically utilizes a metal sheet or foil (such as lithium metal) as the counter electrode, a full cell is composed of two active electrodes—one functioning as the cathode and the other as the anode. The design and assembly of a full cell require the consideration of multiple factors, including the selection of