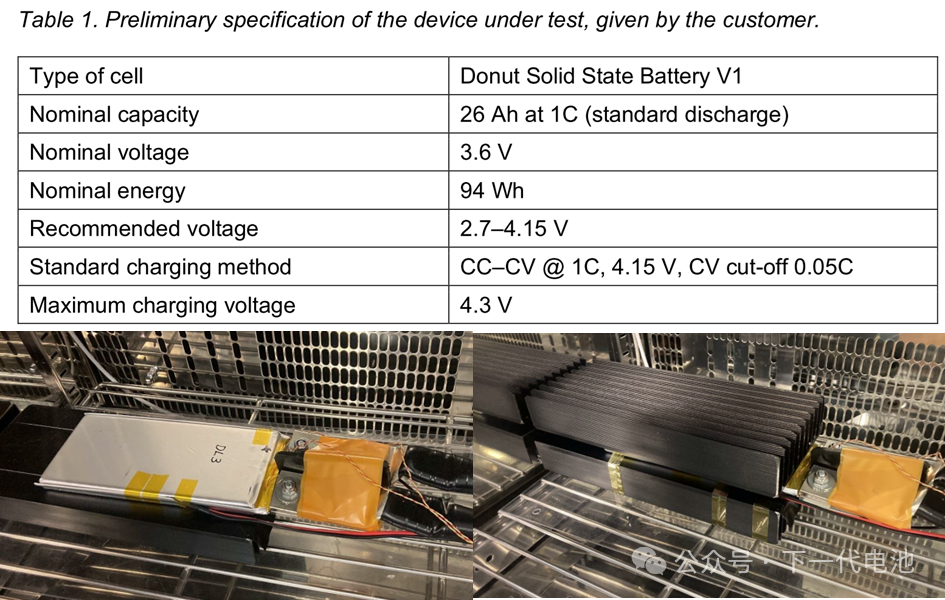
How to map the Neware aux testing channels? 2026 update
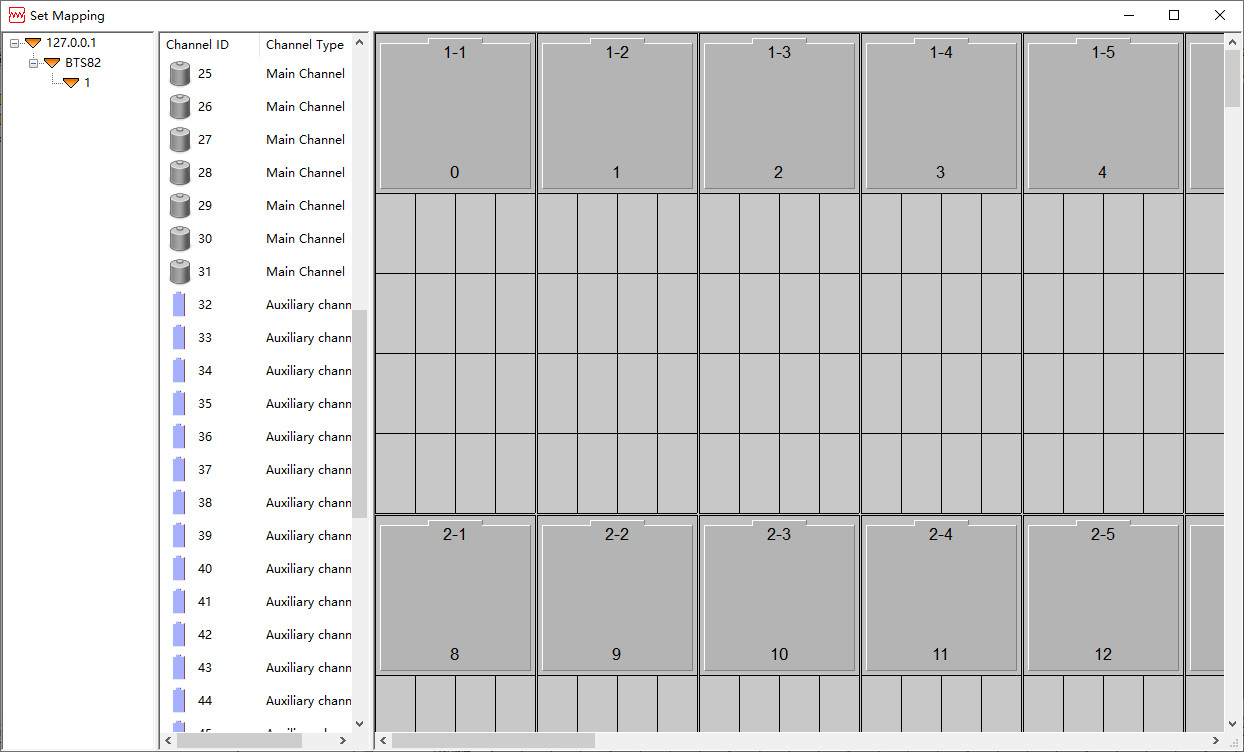
How to map the Neware aux testing channels? This is the latest version of the Neware aux channel; other older versions are still mapped here. The mapping method and location remain largely unchanged. The below tutorial will guide you how to map Neware aux testing channles in software. Open the Set-Mapping window, you can find the blue battery symbol if you have aux channels. (The blue means the aux channel can be mapped. You can not do the aux channel mapping if the aux channels’ color shows grey. ) Click link for “Neware BTS4000 Set up” to learn about how to open the Set-Mapping window. Below is the Set-Mapping window