Introduction to lithium-sulfur battery and lithium-sulfur electrolyte Published in 2026
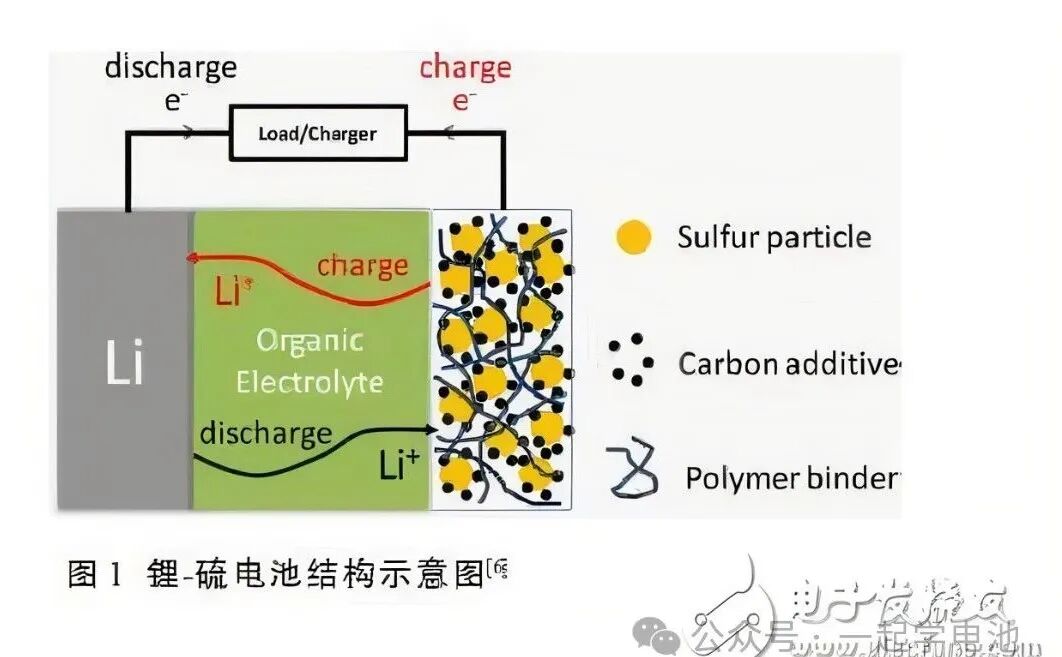
Introduction to lithium-sulfur battery and lithium-sulfur electrolyte Source: WeChat Official Account “Learn Batteries Together” Lithium-sulfur battery (Li-S battery) is a type of lithium battery that uses sulfur as the positive electrode (elemental sulfur is abundant, inexpensive, and environmentally friendly) and metallic lithium as the negative electrode. Due to its high energy density and low-cost raw materials, it is considered a potential candidate for next-generation high-performance batteries. The electrolyte, as a crucial component of lithium-sulfur batteries, directly affects the battery’s performance and lifespan. Note: The positive electrode material of lithium-sulfur batteries is generally composed of sulfur and a highly conductive material (sulfur itself is non-conductive, so a conductive agent, and a