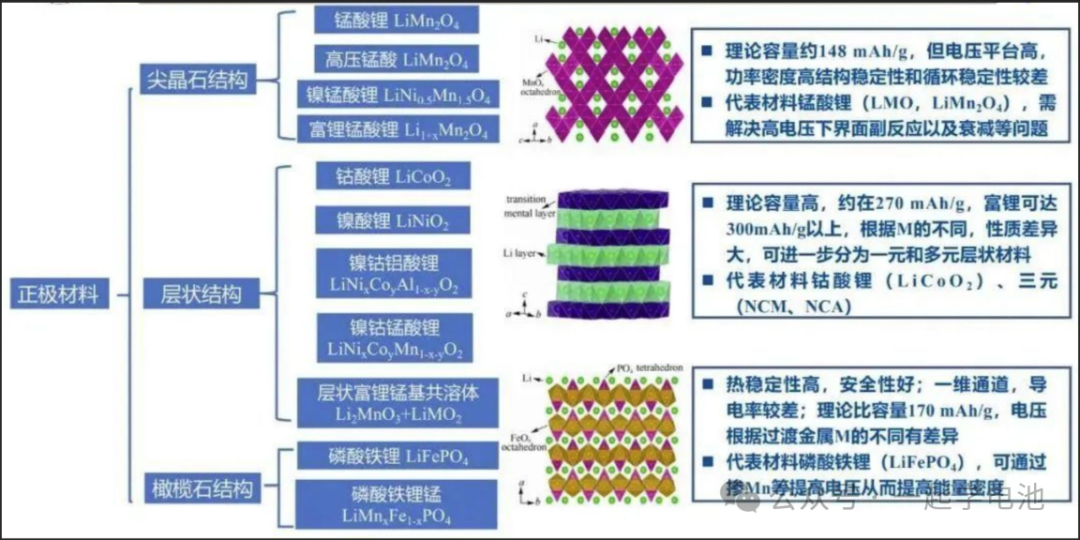
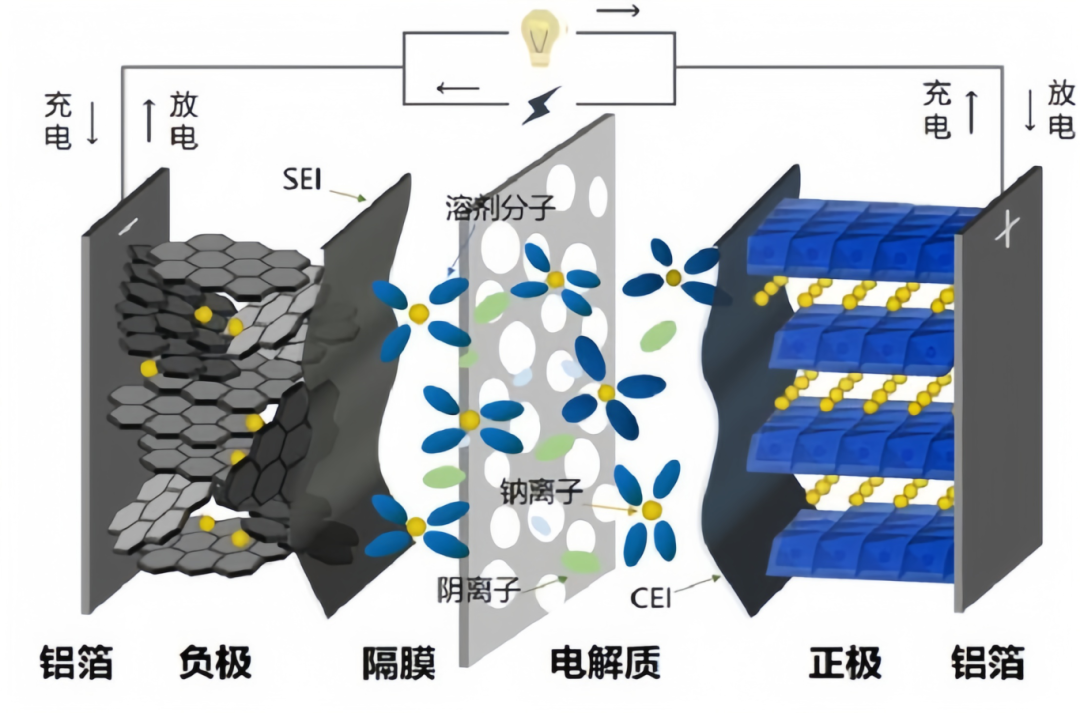
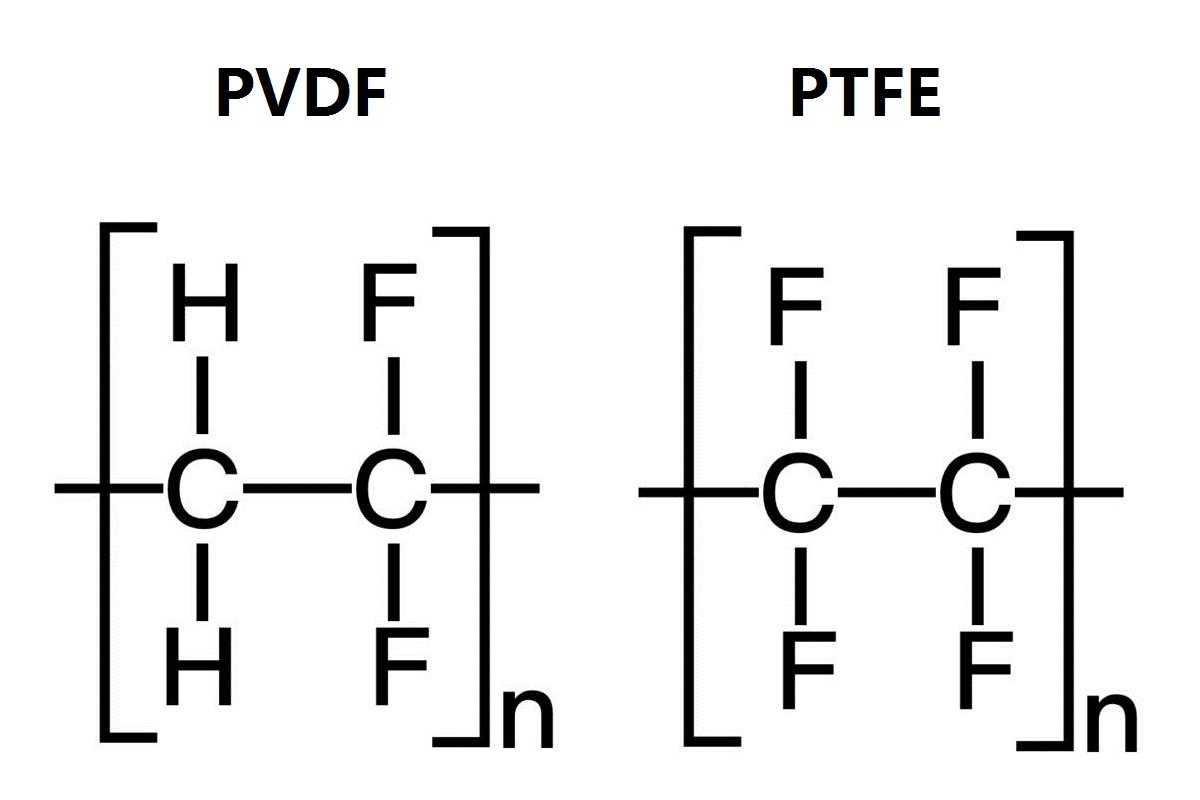
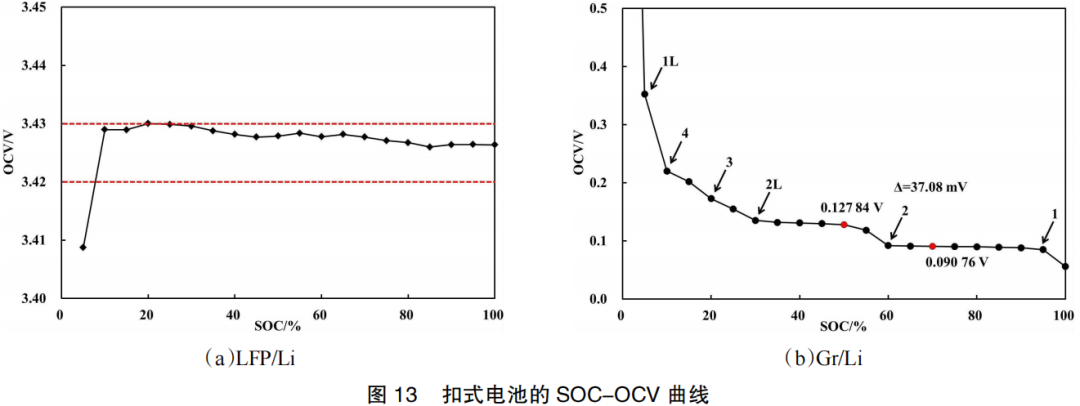
Lithium Plating in Lithium-ion Batteries: Causes and Manifestations 2026
Lithium Plating in Lithium-ion Batteries: Causes and Manifestations I. Why Does Lithium Plating Occur? In simple terms, when the anode cannot promptly accommodate or intercalcate lithium ions migrating from the cathode, these ions “pile up” on the surface of the anode and are reduced into metallic lithium. As this deposited lithium continues to accumulate, it evolves into lithium dendrites. These dendrites typically manifest in four distinct morphologies: whisker-like, mossy, dendritic (tree-like), and spherical. The main causes can be categorized into two major groups: operating conditions and battery states. 1. Operating Conditions (External Factors) Low-Temperature Charging: This is one of the most common causes. At low temperatures: The diffusion rate